FDA updates ByHeart botulism outbreak – releases inspection reports

The FDA and CDC, in collaboration with the California Department of Public Health (CDPH), the Infant Botulism Treatment and Prevention Program (IBTPP), and other state and local partners, continue to investigate an outbreak of infant botulism in several states. Epidemiological and laboratory data show that ByHeart Whole Nutrition infant formula may be contaminated with Clostridium botulinumwhich causes childhood illnesses in several regions of the country.
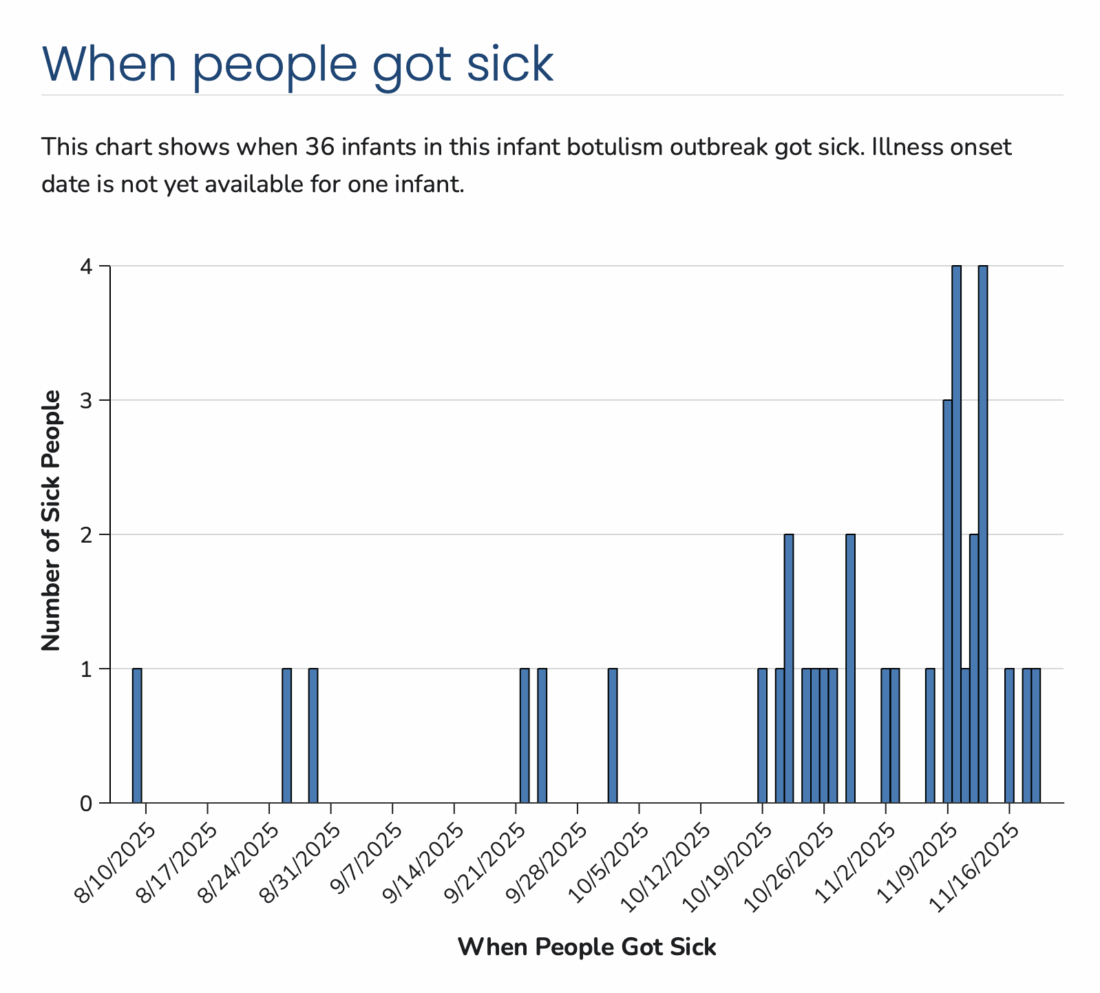
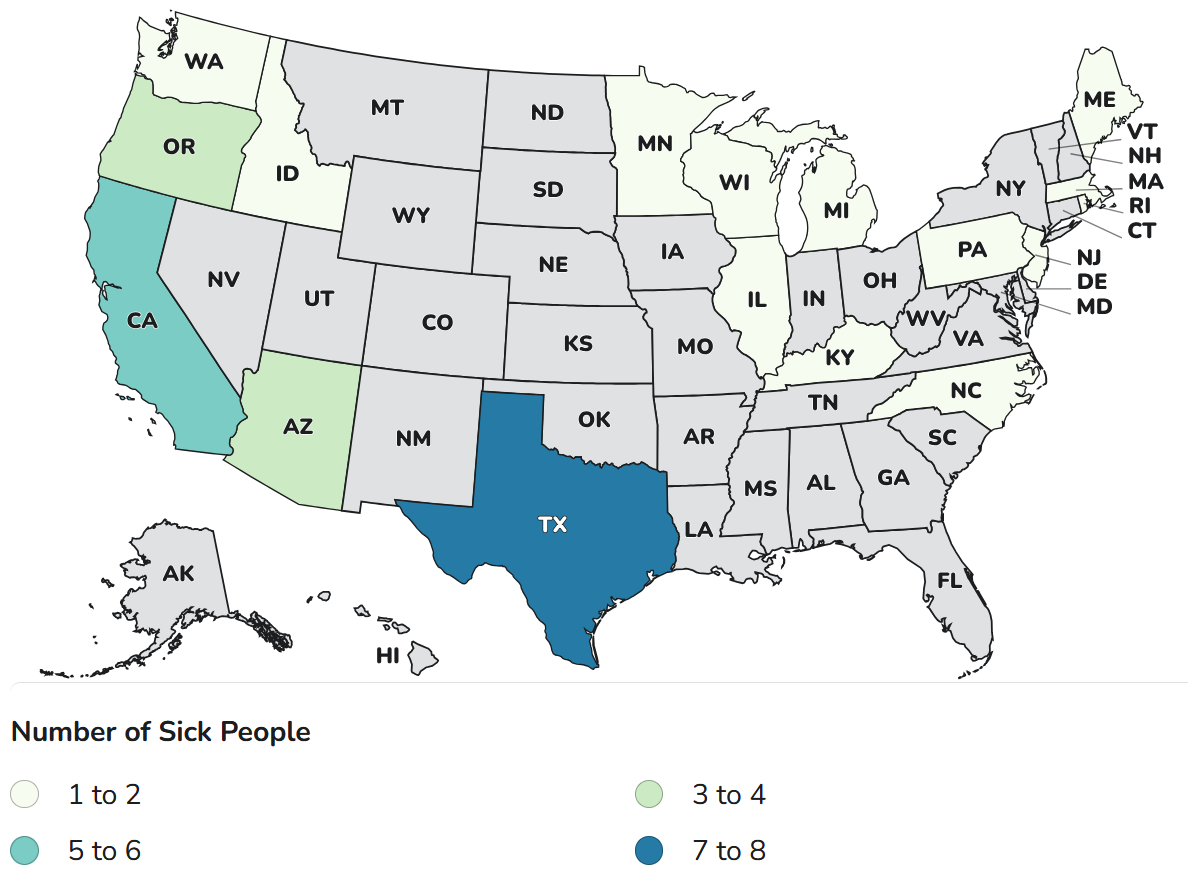
As of November 26, 2025, a total of 37 infants with suspected or confirmed infant botulism and confirmed exposure to ByHeart Whole Nutrition infant formula (various lots) have been reported in 17 states: Arizona 3, California 5, Idaho 1, Illinois 2, Kentucky 1, Massachusetts 2, Maine 1, Michigan 1, Minnesota 2, North Carolina 2, New Jersey 1, Oregon 3, Pennsylvania 1, Rhode. Island 1, Texas 8, Washington 2, and Wisconsin 1. Laboratory confirmation of some cases is underway. For the 36 cases for which information on illness onset is available, the illnesses began on dates ranging from August 9 to November 19, 2025. All 37 infants were hospitalized. No deaths have been reported to date. For the 35 infants for whom age and sex information is available, they range in age from 16 to 264 days and 15 (43%) are girls.
State and local public health officials ask caregivers about the foods infants were fed in the month before they became ill. Thirty-seven infants were identified and were fed ByHeart Whole Nutrition powdered formula before becoming ill.
The FDA has received reports that the recalled formula is still on store shelves in several states, including at several Walmart, Target, and Kroger locations, and at one or more Sprouts Organic Market, Safeway, Acme, Jewel-Osco, Shaw’s, Star Market, Smith’s, King Sooper’s, Albertson’s, Whole Foods, Wegman’s, and Publix locations. The FDA is working with state partners and retailers to ensure an effective recall and immediate removal of these products from store shelves across the country. All ByHeart infant formula products have been recalled and these products are not expected to be available for sale in stores or online. This includes all “anywhere” single-serve cans and sticks.
Additional testing by ByHeart, FDA, CDC and state partners is underway and results are expected in the coming weeks.
The FDA investigation, including on-site inspections, is ongoing to determine the point of contamination. The FDA publishes Establishment Inspection Reports (EIRs) and FDA Form 483 inspection observations from completed inspections previously conducted between 2022 and March 2025 at ByHeart facilities. ByHeart, Inc. is the parent company of three Blendhouse manufacturing plants in Allerton, Iowa; Portland, Oregon; and Reading, Pennsylvania.
The FDA investigation is ongoing to determine the point of contamination. This notice will be updated as information becomes available.
Number of cases
Total illnesses: 37 (6 new)
Hospitalizations: 37 (6 new)
Deaths: 0
Last appearance of the disease: November 19, 2025
States with cases: AZ, CA, ID, IL, KY, MA, ME, MI, MN, NC, NJ, OR, PA, RI, TX, WA, WI
Product Distribution: Online and Nationwide (including Guam and Puerto Rico) and Internationally
Useful links
Product images


Example results
Product sampling and testing is conducted by the FDA, CDC, state partners and ByHeart. Available information on positive samples is included below. This table will be updated as additional results become available or shared with the FDA.
Due to the large number of samples, only positive results are reported here. The detection of Clostridium botulinum in infant formula is complex and a negative test result does not exclude the presence of the bacteria in the product.
Parents and guardians should not use any ByHeart infant formula regardless of test results.
| Sample collected/analyzed by | Product | Test result | Type of toxin |
|---|---|---|---|
| CDPH | ByHeart Infant Formula Box (Lot #251131P2) | Positive | Type A |
| By heart | ByHeart Infant Formula (lots/lots not reported) | Positive | Type A |
International distribution
The ByHeart infant formula recall impacts markets outside the United States. Customer information provided by Amazon shows that a limited quantity of recalled ByHeart infant formula was distributed in Argentina, Brazil, Brunei, Canada, Chile, China, Colombia, Ecuador, Egypt, Hong Kong, Israel, Jamaica, Japan, Republic of Korea, Peru, Philippines, Romania, Singapore, South Africa, Thailand and the British Virgin Islands.
Consumers worldwide should not use any ByHeart brand infant formula as all ByHeart products are included in this recall.


