Number of patients in outbreak traced to moringa leaf products growing fast

Twenty more people have been confirmed as patients in a salmonella outbreak linked to moringa leaf capsules and powder.
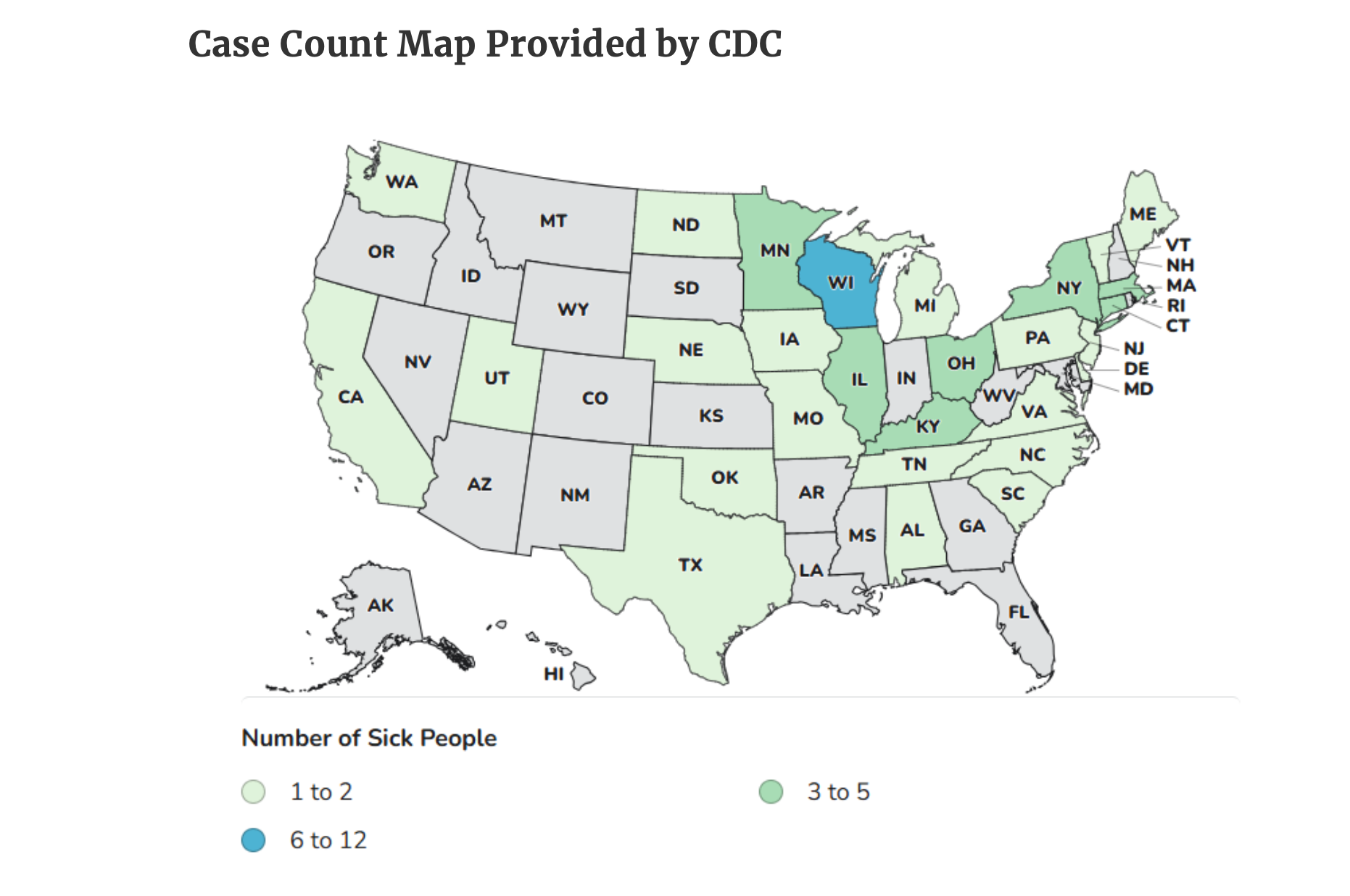
Since the last update from the Centers for Disease Control and Prevention on January 15, the number of sick people has increased to 65, at least 14 of whom have required hospitalization. No deaths have been reported.
The outbreak of Salmonella Typhimurium and Salmonella Newport infections is linked to the recall of Why Not Natural Pure Organic Moringa Green Superfood capsules and the recall of Live it Up brand Super Greens dietary supplement powder, according to the Food and Drug Administration.
The sick stretch from coast to coast and span 28 states. The illnesses began on dates ranging from August 22, 2025 to January 11, 2026.
The actual number of sick people in this outbreak is likely much higher than the number reported, and this outbreak may not be limited to states with known illnesses. This is because many people recover without medical care and others are not specifically tested for salmonella. Additionally, recent illnesses may not yet be reported because it typically takes three to four weeks to determine whether a sick person is part of an outbreak. The CDC estimates that for every confirmed patient in a salmonella outbreak, 29 go undetected.
Of the 40 people surveyed so far, 35 reported eating a product containing moringa leaf powder, with 31 reporting only Live it Up Super Greens supplement powders, three reporting only Why Not Natural moringa powder capsules, and one person reporting eating both products.
The FDA’s traceability investigation revealed a common manufacturer between Live it Up brand Super Greens capsules and Why Not Natural Pure Organic Moringa Green Superfood capsules that used moringa leaf powder in both products. The FDA has not named the manufacturer.
On January 24, Art Monkey LLC, trading as Why Not Natural of Houston, Texas, discontinued the sale of Pure Organic Moringa Green Superfood capsules and launched a reminder from batch #A25G051 on January 28. The FDA is working with companies to determine the root cause of the contamination.
On January 20, Superfoods Inc. expanded its recall of Live it Up brand Super Greens product to include domestic distribution of the recalled product, including Puerto Rico, Guam and the U.S. Virgin Islands, as well as international distribution to consumers in the United Kingdom. On January 26, 2026, the Superfoods, Inc. recall press was updated to make changes to the net weight of recalled Live it Up brand Super Greens Wild Berry products. See the recall notice for more information.


