Can a groundbreaking cancer therapy help people with multiple sclerosis?

Grace Miller was a 24-year-old law school student when she was diagnosed with multiple sclerosis.
For years prior, she’d had severe fatigue, which a neurologist initially suspected was caused by narcolepsy. When she was struck with bouts of vertigo and eventually temporary vision loss, the doctor determined the correct diagnosis.
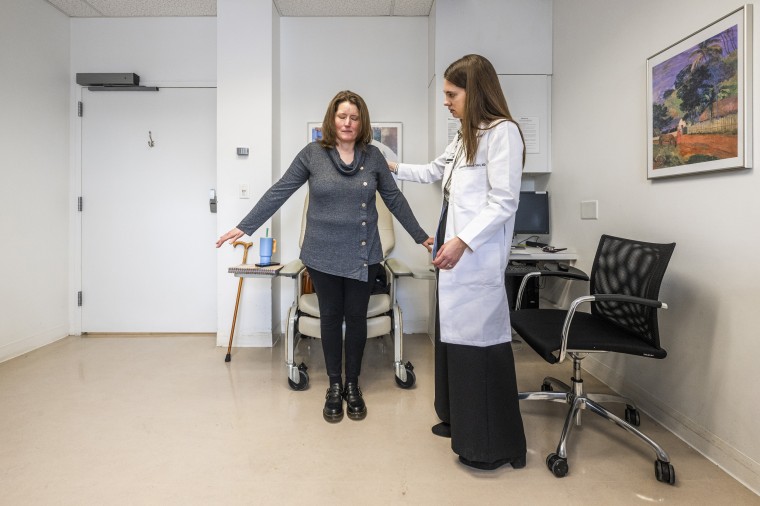
Miller, now 46, of Fishers, Indiana, spent 15 years on two different medications that made her feel sick every time she got a new dose. Her vision has faded in and out over the years, and in 2021, the mother of two started walking with a cane.
“I used to work as a lawyer, but reading a book is not even an option anymore,” she said.
Then, last year, Miller enrolled in an early-stage clinical trial at the Cleveland Clinic for a cancer therapy that researchers across the world are hoping could halt MS.
That treatment, called CAR-T therapy, had been a groundbreaking step forward for cancer patients.
During CAR-T therapy, doctors extract a person’s own T cells — immune cells that usually fight infection — and reprogram them to identify and attack proteins found on tumor cells. When the T cells are infused back into the person’s body, they become cancer-fighting cells. The treatment has proved to be particularly effective against cancers that affect a different immune cell, the B cell. These include types of leukemia and lymphoma.
Rogue B cells also play a major role in multiple sclerosis. The cells become overactive and attack myelin, a fatty substance that protects and insulates nerve fibers in the central nervous system. Without myelin, signals that would normally rapidly fire across nerves are slowed or blocked. Although there are treatments that can stave off progression, there is no cure for MS. Over time, people with MS can lose function in their limbs, lose their vision and develop memory problems.
There are reasons to believe CAR-T therapy may be able to target the same overactive B cells that current multiple sclerosis drugs already do. The difference is that the current drugs cannot get to B cells in the brain and central nervous system, but CAR-T cells appear to be able to.
“Not only would they kill B cells in the blood, like our already approved antibody therapies do, but also the ones that are hiding in the brain, which we think play an important part in progression,” said Dr. Jeffrey Cohen, director of the experimental therapeutics program at the Cleveland Clinic’s Mellen Center for Multiple Sclerosis. Inflammation caused by dysregulated B cells in the brain and central nervous system makes MS symptoms worse, he added.
“The fact that there is already pretty extensive experience with this CAR-T approach in cancer gives us a big head start,” said Cohen, who is leading the Cleveland Clinic trial. He emphasized that all CAR-T trials for MS are very early exploratory studies, and it is still unclear whether the treatment will be successful.
Similar trials are underway across the country, including ones at Stanford University, Mass General and Columbia University. The Cleveland Clinic trial — sponsored by the drug company Bristol Myers Squibb — has recruited four people with MS as well as three people with another autoimmune disease called myasthenia gravis. Two of the people with MS, including Miller, have progressive MS, a form of the disease marked by gradual worsening of neurological function and disabilities over time. The other two have a more common form of MS, called relapsing MS. In these cases, people have unpredictable attacks and temporary relapses followed by a period of recovery.
CAR-T typically only requires one infusion of modified immune cells. (Instead of modifying the cells to target tumor cells, in this case, they’re reprogrammed to attack the overactive B cells.) After a person’s T cells are removed, they undergo a round of chemotherapy to make space for the modified cells that will be infused back into their body. All four people in the trial who have MS received their infusion between six and 12 months ago.
Miller got hers last May. Ten months out, she still walks with a cane — Cohen told her they can’t fully count progress until she can walk without it — but is increasingly taking steps on her own.
“My friend brought her 18-month-old to my house this fall and I picked her up for the first time standing up. I have never done that before,” Miller said.
Working toward regeneration
All the CAR-T trials for MS are in extremely early stages. Researchers are exploring whether the approach could work, and Cohen is careful to caution that it may not. Some experts are not convinced that the therapy will be meaningfully more effective than existing treatments.
“The antibodies that we have do a good job, even though they don’t penetrate the blood-brain barrier,” said Dr. Rhonda Voskuhl, director of the UCLA Multiple Sclerosis Program, who is not involved in any of the CAR-T MS trials.
Voskuhl said she is hesitant to believe CAR-T will be an effective treatment for people who have progressive MS in particular. In these cases, the number of new lesions in the brain caused by overactive B cells — which cause the disabilities seen in people with the disease — slows, and much of the damage is done, she said.
“I don’t think you can anti-inflammatory yourself out of MS when it’s both an inflammatory and neurodegenerative disease,” she said. “We also need something that is going to bind to brain cells and protect and heal them.”
Dr. Enrique Alvarez, medical director of the Rocky Mountain MS Center at CU Anschutz in Aurora, Colorado, agreed there is a need for therapies that repair neurological damage in people with MS. That would require effectively growing new neural pathways and reversing cell damage. The most promising way to do that is using stem cells.
“Nobody is going to argue with the need for something that can repair what is already broken, but right now, I’m not sure that stem cells are at a point where we are getting great success in it,” said Alvarez, who is also not involved in any CAR-T MS trial.
In the meantime, Alvarez said, improving upon existing MS drugs is a worthwhile venture. One MS drug, a B cell immunotherapy called ofatumumab, was originally developed for B cell leukemia.
“We don’t know if CAR-T could stop inflammatory disease, but if we could prevent people from needing further treatments for the rest of their lives, that’s worth looking into,” he said.
Cohen said he doesn’t believe that CAR-T cells will be able to repair damage caused by MS, and agreed that therapies that can do that are badly needed. The body can repair some of this damage on its own, he added.
“The problem is, MS causes the damage more quickly than the body can repair. If we can inhibit the MS damage, the repair can maybe catch up,” Cohen said.
CAR-T is not without risk. The treatment can cause a serious reaction called cytokine release syndrome, in which the immune system releases a storm of inflammatory proteins into the bloodstream. It can cause flulike symptoms that can progress into organ failure. Another potential risk is immune effector cell-associated neurotoxicity syndrome, or ICANS, which causes an inflammatory reaction in the central nervous system. Cohen said ICANS is a particular concern in people with MS.
Another concern is that CAR-T therapy could prove riskier in MS patients than researchers thought, Cohen said.
“There could be complications we didn’t anticipate because cancer patients are different from MS patients,” Cohen said. “It could turn out to not be effective, so we have to be cautious at this stage.”
CAR-T technology is evolving quickly and has buy-in from many of the world’s largest drug companies, meaning the therapy is constantly being fine-tuned in ways that could make the modified T cells better at targeting dysregulated B cells. The trials will also give a more clear answer as to whether targeting B cells in the central nervous system makes a difference in MS disease progression, Alvarez said.
“Even if CAR-T doesn’t work, the information we get about the immune system and MS from these trials will be important,” he said.




