Precise, minimally evolved adenine base editors generated through mutation reversion analysis

Activity of ABE7.10 single-residue reversion variants at episomal reporter and endogenous sites identifies both essential and dispensable mutations
We previously reported that reversion of the single D108N mutation in ABE7.10 (to produce the editor that we call ABE7.10-N108D) resulted in an average 146-fold decrease in editing activity, as measured across 6 endogenous genomic sites in mammalian cells19. This suggests that the D108N mutation introduced in the first round of directed evolution of ABE7.10 was critical for both enhancing DNA editing activity by TadA and maintaining this activity even in the context of the 13 other acquired mutations. To investigate how the additional mutations in ABE7.10 impact base editing activity, we generated all 14 single-residue ABE7.10 reversion editors and evaluated their activities using a previously reported set of 8 episomal, highly sensitive, fluorescence turn-on reporters (Fig. 2a and Supplementary Discussion 1)12.
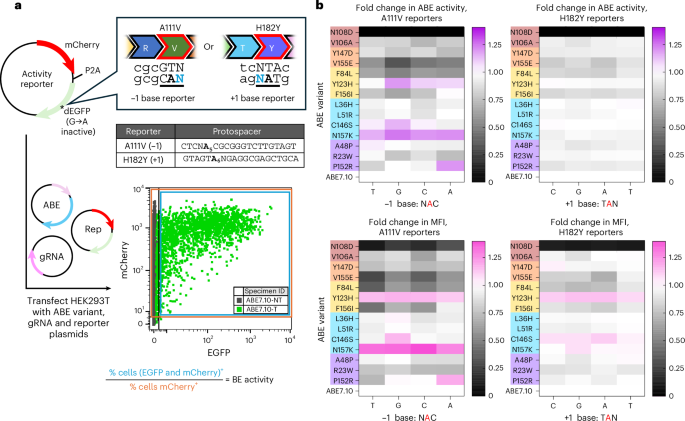
a, BE activity was assessed using a series of fluorescent turn-on plasmid reporters. Each reporter plasmid (Rep) coded for a transfection marker mCherry fluorescent protein linked to EGFP reporter protein via a P2A ribosomal skipping sequence. The deactivated EGFP reporter protein (dEGFP) required an A•T to G•C correction at the DNA level to restore EGFP fluorescence. The target A was placed such that the base either directly 5′ (−1) or 3′ (+1) fell at a wobble codon position, allowing for substitution of any base N at this position without impacting the resulting protein sequence and fluorescence. For the −1 family of reporters, the target A fell at position 5 while in the +1 family of reporters the target A was at position 6. HEK293Ts were transfected with Rep, base editor plasmid and appropriate gRNA and incubated for 72 h, after which EGFP fluorescence turn-on was evaluated using flow cytometry. b, ABE7.10 single-residue reversion editors were evaluated for editing activity by calculating their fold change in activity relative to that of ABE7.10. Relative activities of reversion editors were determined using BE activity scores (top set of heat maps) and the MFI of the double-color (mCherry and EGFP)+ cell population (bottom set of heat maps). Data are separated by reporter family, with A111V (−1 base reporters) on the left and H182Y (+1 base reporters) on the right. Heat map scales have been set so white indicates equivalent activity to ABE7.10, gray scale indicates activity less than ABE7.10 and color indicates activity greater than ABE7.10. Editor variants are ordered by round of evolution their mutation was introduced in, with color coding adherent to Fig. 1. Values represent the average of fold changes calculated for n = 3 independent biological replicates.
Source data
We performed our reversion analyses using monomeric forms of ABE (TadA*-nCas9, where TadA* indicates a mutated TadA variant). While wild-type TadA is known to act as a dimer on its native tRNAArg substrate in E. coli11,20, and the crystal structure of ABE8e captures TadA* in a homodimeric form15, many researchers have found no significant difference in editing activity when delivering monomeric versus dimeric ABE7.10 and ABE8 variants12,14,19,21. HEK293T cells were transfected with a mixture containing one of the eight reporter plasmids, its respective gRNA plasmid and a monomeric base editor plasmid encoding one of the 14 ABE7.10 single-residue reversion editors or ABE7.10. Cells were incubated for 72 h to allow for editing and fluorescent protein expression before analysis by flow cytometry (Supplementary Fig. 2). Cells were analyzed for both mCherry and EGFP fluorescence, and a ‘BE activity’ score was calculated by dividing the percentage of edited cells by the total percentage of transfected cells (Fig. 2a). While this BE activity score is not equivalent to editing efficiency, it allows for comparisons of relative editing activities among multiple BE variants and helps account for variation resulting from differences in transfection efficiency12 (Supplementary Figs. 3a,b, 4 and 5). The median EGFP fluorescence intensity (MFI) of double-color-positive cells was used as a secondary marker of BE activity (Supplementary Figs. 3c,d, 6 and 7).
All BE activity scores and MFIs were normalized to that of ABE7.10 within a given biological replicate to enable fold-change comparisons of how each reversion variant impacted activity relative to ABE7.10 (Fig. 2b and Supplementary Fig. 8). In general, we observed more drastic changes in relative activities of the reversion variants with the A111V (−1) family of reporters compared with the H182Y (+1) family of reporters. This is consistent with previous studies that show the identity of the −1 base to have a much stronger impact on relative ABE editing efficiency12,22. While the H182Y reporters showed a smaller dynamic range, they still captured a dramatic reduction in activity for the ABE7.10-N108D variant, and significant differences in activity between several reversion editors and ABE7.10 (Fig. 2b and Supplementary Figs. 5 and 7). Overall, this suggests that these two families of reporters can detect changes in editing activity across diverse sequence contexts.
The data from all 14 ABE7.10 single-residue reversion editors tested against all eight fluorescent turn-on reporter plasmids are shown in Fig. 2b and Supplementary Figs. 3–9. Notably, the ABE7.10-N108D reversion editor had the greatest impact on editing activity for all reporters, showing average normalized activity scores relative to the parental ABE7.10 editor (1.00 ± 0.10) of (2.07 ± 0.95) × 10−4 for the −1 family of reporters and (1.04 ± 0.31) × 10−3 for the +1 family of reporters. Similar extreme decreases in editing activity for the ABE7.10-N108D variant were reflected with MFI values (Fig. 2b and Supplementary Figs. 3c,d and 8b). While no other single-residue reversion variant had as deleterious an impact on editing activity as N108D, the reversions V155E, F84L and F156I also resulted in significantly decreased editing activity across all reporters. We observed average normalized activity scores relative to ABE7.10 of 0.66 ± 0.05, 0.74 ± 0.06 and 0.85 ± 0.07 across all eight reporters for the V155E, F84L and F156I reversion variants, respectively. The V106A, Y147D and R23W reversion editors also reduced editing activity but to a lesser degree, with average activity scores relative to ABE7.10 of 0.88 ± 0.08, 0.89 ± 0.08 and 0.93 ± 0.09, respectively, across all eight reporters.
From these data, we also identified five reversions (C146S, L36H, A48P, P152R and L51R) that had minimal impact on editing efficiency (for example, an overall normalized average base editing activity score between 0.95 and 0.99 relative to ABE7.10), regardless of sequence motif (Supplementary Fig. 8). Finally, we found two reversion variants (Y123H and N157K) that increased activity scores relative to ABE7.10, an effect that was more apparent with the −1 reporters (Fig. 2b and Supplementary Fig. 8). ABE7.10-Y123H had an increased BE activity score relative to ABE7.10 of 1.10 ± 0.20 with the −1 reporters and 1.04 ± 0.10 over all reporters, while ABE7.10-N157K had an increased BE activity score relative to ABE7.10 of 1.21 ± 0.23 with the −1 reporters and 1.09 ± 0.12 overall. Trends in MFI changes mirrored those of the activity scores (Fig. 2b and Supplementary Figs. 3, 8 and 9).
These data allowed for some assessment of editor sequence motif preference, with a greater focus placed on the identity of the base 5′ of the target A. Generally, we noted that the sequence motif preferences of most single-residue reversion editors matched that of ABE7.10: TAC > CAC > AAC ~ GAC (Supplementary Fig. 10 and Supplementary Discussion 2). These data suggested that no reversion strongly favored any one sequence motif over others. Taken together, these data identified that certain mutations accumulated during the directed evolution of ABE7.10 remained necessary for base editing activity (most crucially D108N and to a lesser degree E155V, L84F and I156F), while others may be dispensable (S146C, H36L, P48A, R152P and R51L) as their reversions had minimal impact on base editing activity across these eight reporters. We also identified two mutations that appear detrimental to base editing activity (H123Y and K157N), as their reversions on average increased editing activity across all eight reporters.
We next evaluated each ABE7.10 single-residue reversion editor for activity at three genomic sites (selected for their varied target motifs) to confirm that the results from the episomal editing assay extended to endogenous editing (Extended Data Fig. 1a,b and Supplementary Fig. 11). The trends in these data were consistent with what we observed for episomal editing (Supplementary Discussion 3). For a subset of reversion variants that altered editing activity relative to ABE7.10, we developed alanine substitution variants and similarly profiled editing activity at these three endogenous sites (Extended Data Fig. 1c–f). While alanine substitution allowed for investigation into biochemical interactions that may drive changes in deaminase activity (Supplementary Figs. 12 and 13, and Supplementary Discussion 4), no alanine substitution consistently improved editing activity at all three sites. Together, these results at endogenous genomic sites support observations from the episomal fluorescent turn-on reporter assay, suggesting that some reversions have minimal impact on or even improve ABE editing activity.
Stability of expression assays shows no drastic decreases in protein expression levels for ABE7.10 single-residue reversion variants
It has been hypothesized that during the course of directed evolution, mutations that increase activity can often recompense for this change with a concurrent loss in protein stability17,23. This hypothesis has not been investigated in cellular contexts for mutations accumulated by TadA during the development of ABEs. In addition, it is hypothesized that some mutations acquired during directed evolution, particularly those distal from an enzyme’s active site, compensate for destabilizing mutations by providing allosteric support or additional stability without directly impacting catalytic activity24,25. While some mutations acquired during the evolution from ABE7.10 into ABE8e show this behavior26, none of the initial mutations in ABE7.10 have previously been identified as compensatory. Reversion of such a compensatory mutation could lead to additional destabilizing effects, impacting the reversion variant’s activity. Given our findings that several ABE7.10 single-residue reversions modified base editing activity, we sought to evaluate whether altered intracellular ABE protein levels could account for these changes in editing activity.
To this end, we adapted a fluorescent reporter assay in which the ratio of EGFP/mCherry fluorescence measured relative intracellular expression levels of the ABE reversion variants in HEK293T cells (Extended Data Fig. 2a and Supplementary Discussion 5)27. Preliminary studies with this assay found diminishing ABE expression for more evolved editors (ABE0.1 > ABE7.10 > ABE8e; Supplementary Fig. 14 and Extended Data Fig. 2b), results that were verified by independent western blot analysis (Extended Data Fig. 2c and Supplementary Discussion 5). We extended the fluorescent reporter assay to examine stability of expression for all 14 ABE7.10 single-residue reversion variants (Extended Data Fig. 2d and Supplementary Fig. 15). Generally, we noted a pattern in which reversion of mutations acquired in earlier rounds of evolution tended to increase expression and stability while reversion of mutations acquired in later rounds of evolution tended to decrease expression and stability (Supplementary Discussion 5). This lends support to the hypothesis that some later mutations may act in a compensatory manner with regard to protein expression and stability. Of note, none of the ABE7.10 single-residue reversion variants that increased editing activity or were deemed neutral in the fluorescent reporter EGFP turn-on assay (indicated with asterisks in Extended Data Fig. 2d) showed significantly increased EGFP/mCherry fluorescence intensity ratios. Some of these single-residue reversion editors were further evaluated for ABE protein expression levels via western blot (using an ABE-P2A-mCherry construct) and were confirmed to show similar expression levels to ABE7.10 (Extended Data Fig. 2c). Taken together, these data indicate that the changes in ABE activity caused by the reversion mutations were not driven by changes in expression and stability, but rather by changes in enzyme activity.
ABE7.10 single-residue reversion variants show disparate behavior in a bacterial host
Although the majority of ABE applications are in mammalian systems, the directed evolution scheme used to develop ABE7.10 occurred in an E. coli bacterial host2. This is notable owing to the possibility of endogenous TadA dimerizing in trans with the evolving TadA of the ABE, an interaction not possible in mammalian hosts that lack a TadA homolog11,15,26,28. Given these complex interactions, as well as additional differences between mammalian and bacterial systems (including presence or absence of chaperone proteins, differences in posttranslational modifications, availability of critical cofactors or differences in DNA repair pathways), we hypothesized that alternate editor activity may be observed between these two hosts29,30,31. We therefore developed an assay to quantify editing activities of the single-residue reversion variants in a bacterial host system that used bacterial survival rate as a measure of ABE activity (Fig. 3a and Supplementary Discussion 6).
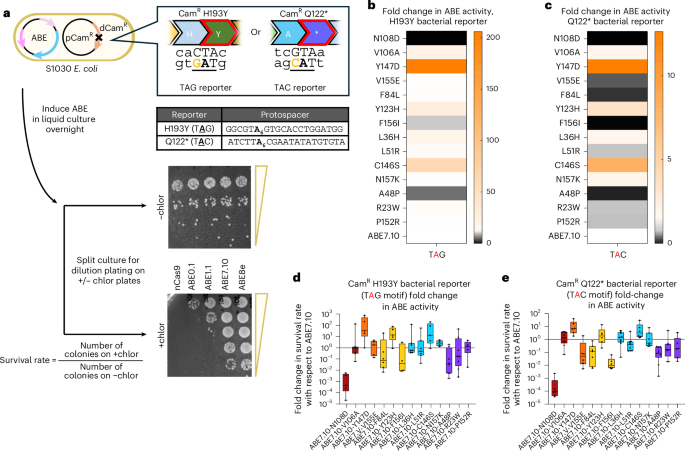
a, Base editing in bacteria was measured using an antibiotic survival assay. Sequence encoding a dead chloramphenicol acetyltransferase (CamR) requiring an A•T to G•C correction to restore antibiotic resistance was placed on a carrier plasmid, pCamR. Two versions of this were developed: one with the deactivating mutation H193Y, where the target A had the sequence motif TA6G, and the other a Q122* mutation where the target A had the sequence motif TA6C. S1030 E. coli harboring pCamR were transformed with various ABE7.10 single-residue reversion plasmids optimized for bacterial editing. ABE plasmids also contained appropriate gRNAs for targeting the mutation in CamR to reactivate chloramphenicol resistance, as well as a theophylline-inducible riboswitch to temporally control expression of the base editor. Bacteria were allowed to grow overnight in liquid culture with the base editor expressed, before splitting the culture and dilution plating each sample on +/− chloramphenicol plates (+/− chlor). Colony counts on +/− chlor plates were used to determine the survival rate of a given single-residue reversion editor in bacteria. b,c, Heat maps showing average fold-change values in survival rate for single-residue reversion editors against the H193Y TAG reporter (b) and Q122* TAC reporter (c). d,e, Fold change in survival rates relative to ABE7.10 for bacterial base editing activity assay with the H193Y TAG (d) and Q122* TAC reporters (e). Fold-change survival rate measurements were calculated for n = 9 plating pairs per sample with ROUT 1% outliers removed, and all remaining fold-change values plotted individually. The box plot center illustrates median fold change, hinges show quartiles and whiskers show minimum and maximum fold-change values.
Source data
For most reversion variants, we observed similar trends in relative editing activity in the bacterial system as in the mammalian episomal and endogenous editing assays (Fig. 3b–e and Supplementary Fig. 16). Specifically, ABE7.10-N108D was found to be the most deleterious reversion variant, showing an activity that was on average four orders of magnitude lower than that of ABE7.10 in both tested reporters, with survival rates on par with that of ABE0.1 (Fig. 3d,e and Supplementary Fig. 16). The three reversion editors that moderately decreased editing in mammalian systems, V155E, F84L and F156I, generally followed these same trends in bacteria. In the Q122* TAC reporter system, all three showed average survival rates two orders of magnitude below that of ABE7.10. For the H193Y TAG reporter, the F84L and F156I reversion variants both lowered average survival rates by an order of magnitude relative to ABE7.10, while the V155E variant’s activity showed no significant difference. Overall, agreement in activity trends across the three base editing activity reporters analyzed suggests critical roles for the N108D, E155V, L84F and I156F mutations independent of the host system or DNA context (episomal versus chromosomal).
The reversion variant with the greatest relative increase in editing activity in bacteria was ABE7.10-Y147D, which decreased editing slightly in both mammalian-based assays (Fig. 2b and Extended Data Fig. 1b), but resulted in an average 200-fold increase in editing activity compared with ABE7.10 in the H193Y TAG bacterial system, and a less drastic 13-fold increase measured by the Q122* TAC reporter (Fig. 3b–e). Notably, this residue was further evolved during the development of both ABE8e and ABE8.20. These variants were both evolved in bacteria (starting from ABE7.10), with the Y147D reversion mutation selected for in ABE8e and the Y147R mutation selected for in ABE8.20 (Supplementary Fig. 1). In light of these data, these mutations may be causing host-cell-specific increases in editing efficiency. Of further interest, neither the ABE7.10-N157K nor ABE7.10-Y123H reversion variants, which caused the largest increases in editing activity in HEK293Ts, had statistically significant impacts in bacteria, resulting in survival rates within error of ABE7.10 (Fig. 3b–e). These host-system-dependent differences in editing activity may explain why some ABE7.10 mutations that were identified during bacterial-directed evolution (such as mutations acquired during evolutionary rounds four and six) did not result in high activity during validation in mammalian systems2. Furthermore, these results highlight a potential pitfall in using a different host system for evolution or engineering efforts than the intended end-use system.
Multi-residue reversion editors increased editing activities at endogenous sites
Given that several reversion variants had a minimal or sometimes beneficial impact on base editing activity, we postulated that combinations of these reversions may lead to a similar or synergistic effect on editing activity. Following preliminary studies on a series of combination reversion variants using an episomal reporter and at a subset of endogenous sites (Extended Data Fig. 3 and Supplementary Fig. 17), we identified a core set of reversions for further investigation (Supplementary Discussion 7). We renamed the set of ABE7.10 multi-residue reversion editors listed in Fig. 4a, collectively calling these ME-ABEs. The ME-ABEs, ABE7.10-N157K and benchmark editors ABE7.10, ABE8e and ABE8.20 were further characterized at a panel of 27 additional endogenous genomic sites in HEK293T cells (Supplementary Figs. 18 and 19). For further discussion on site selection and normalization of ABE activity data, see Supplementary Discussion 8. For this set of editors, the average normalized editing values across all sites as a function of position in the protospacer are presented in Fig. 4b, Extended Data Fig. 4 and Supplementary Fig. 20. Using our metric for editing window (Supplementary Discussion 8), we found ABE7.10 and all five ABE7.10 reversion variants to robustly edit between positions 4 and 7, ABE8.20 between positions 3 and 8, and ABE8e between positions 3 and 11.
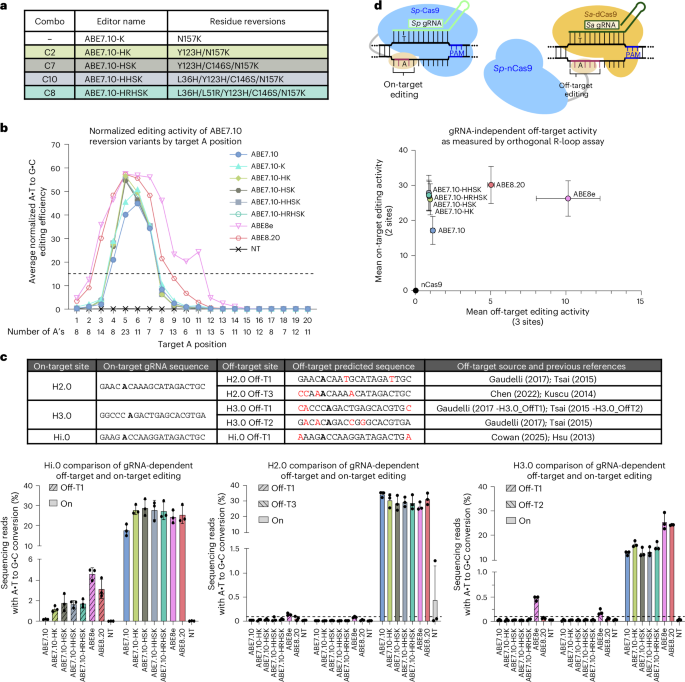
a, Renaming legend for a set of ABE7.10 reversion editors. HEK293T cells were transfected with a single ABE plasmid containing either one of the editors listed, ABE7.10, ABE8e or ABE8.20, along with 1 of 27 gRNA plasmids targeting an endogenous site or an NT control (Supplementary Fig. 18). After a 72-h incubation, genomic DNA was collected, target sites were amplified and sequences were analyzed by NGS. Absolute editing values (percentage of reads with a given A•T pair converted to G•C) were normalized to the absolute editing of ABE7.10 at the H2.0 A5 site (the maximally edited A in all experiments) within a given replicate. Editing activity was examined in n = 3 biological replicates for all ABE7.10 reversion editors, and n = 6 for ABE7.10, ABE8e and ABE8.20. b, Normalized editing scores for all 205 A’s were sorted by position and further averaged across all gRNAs to identify editing windows. The number of A’s considered for each position is given below the x-axis. The horizontal dotted line shows the 15% cutoff used to determine editing windows. c, Sequences of all on-target gRNAs and their predicted gRNA-dependent DNA off-target sites. The most editable A base is shown in bold. Mismatches between on-target and off-target site are shown in red. HEK293T cells were transfected with plasmids encoding each of the ABE variants indicated and an on-target gRNA. Both the on-target and off-target loci were amplified and evaluated for A•T to G•C editing with NGS 72 h after transfection. Bars show average editing measured for n = 3 biological replicates, with error bars showing standard deviation. Dashed horizontal lines indicate 0.1% editing activity, demarcating the limit of reliable detection. d, The orthogonal R-loop assay compares ME-ABE gRNA-independent off-target activity with canonical editors. Briefly, this assay uses two orthogonal Cas constructs; Sp-Cas9 ABE and Sp-gRNA edit an on-target site, while Sa-dCas9 and Sa-gRNA expose an off-target ssDNA R-loop for errant deamination (cartoon, above). HEK293T cells were transfected with plasmids encoding an ABE, an on-target Sp-gRNA, Sa-dCas9, and an off-target Sa-gRNA. After 72 h, genomic DNA was collected and on-target and off-target loci were sequenced. The x–y plot indicates the average off-target activity for the most edited A across three independent off-target sites, and average on-target activity at the most edited A across two independent on-target sites, accounting for n = 3 biological replicates with error bars showing propagated standard deviation.
Source data
When comparing average normalized A•T to G•C editing efficiencies at each position within the editing window, all ME-ABEs were nearly equivalent to one another. They showed notable increases in activity compared with ABE7.10 at positions 4 and 5 while performing similar to ABE7.10 at positions 6 and 7 (Fig. 4b and Supplementary Fig. 20). Of particular note at position 5 (the most highly edited position), the single-residue reversion variant ABE7.10-K had a 1.14-fold increase in editing activity over ABE7.10, while the dual-residue reversion variant ABE7.10-HK and the maximally reverted ABE7.10-HRHSK variant showed 1.41-fold and 1.36-fold increases in editing activity compared with ABE7.10, respectively. Furthermore, all four ME-ABEs on average performed similarly to ABE8e and ABE8.20 at position 5 (ABE8e was only 1.01-fold more active than ABE7.10-HK). Overall, the ME-ABEs show identically narrow editing windows to ABE7.10 and provide an editing efficiency advantage over ABE7.10 at positions 4 and 5. It was previously hypothesized that increases in editing activity by highly evolved ABE variants were always accompanied by an increase in editing window size16. While this is the case when comparing ABE7.10 against ABE8e and ABE8.20, the ME-ABEs decouple this behavior, increasing editing activity while maintaining a narrow editing window.
This series of endogenous target sites also allowed for analysis of editor sequence motif preference (Extended Data Fig. 5, Supplementary Fig. 21 and Supplementary Discussion9). When considering the subset of editable A’s (A’s present in the editing window for a given ABE variant), we found that at GA, AA, TA, AA, AT and AC motifs, the ME-ABEs had average editing activities similar to or higher than that of the ABE8 variants. ME-ABE editing efficiencies were improved with respect to ABE7.10 at CA and AG motifs, but were lower than the ABE8 variants in these sequence contexts. We also use this set of endogenous target sites to look for undesired editing outcomes. Given previous findings that highly active TadA variants can deaminate bystander cytosines within the editing window32, we analyzed these data for cytosine editing. While we noted slightly higher levels of C•G to T•A editing for ABE8e and ABE8.20 compared with ME-ABEs and ABE7.10, global cytosine editing for all editors was within error of non-target controls (Supplementary Figs. 22 and 23 and Supplementary Discussion10). A similar analysis of indels across all 27 sites found median indel rates for all editors to be below 0.3%, with ME-ABEs facilitating indel rates comparable to that of ABE7.10 and slightly lower than those of ABE8e and ABE8.20 (Supplementary Figs. 24 and 25).
Altogether, the four ME-ABEs behaved quite similarly to each other. They maintained the same narrow editing window as ABE7.10 (positions 4–7), but improved editing efficiencies compared with ABE7.10 at most sequence motifs. Measuring the activity of the single-residue reversion editor ABE7.10-K allowed us to see that alone, the N157K mutation resulted in activity between that of ABE7.10 and the multi-residue reversion variants. This supported our hypothesis that the Y123H and N157K reversions combined may be responsible for the improved editing activities of the ME-ABEs and the modification in sequence context specific activity, while the additional L36H, L51R and C146S reversions appeared to have a neutral impact on editing activity.
ME-ABEs are evaluated for off-target activity
Previous efforts to evolve editors from ABE7.10 for enhanced on-target activity have co-evolved increased gRNA-dependent and gRNA-independent off-target editing activity13. Furthermore, TadA-derived editors maintain the ability to deaminate RNA transcriptome-wide16,33. While certain mutations have been found to reduce this off-target activity, it is often accompanied by a reduction in on-target editing from the parent-derived editor13,21,34. We therefore sought to assess whether our ME-ABEs, which showed improved on-target editing activities relative to ABE7.10, also increased their off-target propensities. To assess gRNA-dependent off-target activity, we selected five previously validated off-target sites associated with three different on-target sites. These potential off-targets were evaluated with all four ME-ABEs and the benchmark editors ABE7.10, ABE8e and ABE8.20 (Fig. 4c and Supplementary Fig. 26)2,14,35,36,37,38,39,40. HEK293T cells were transfected with plasmids encoding an ABE variant and one of the three gRNAs (or a non-targeting control). Cells were incubated for 72 h before genomic DNA was collected and the on-target and relevant off-target site(s) were amplified and sequenced. ME-ABE on-target editing was within error of ABE8e and ABE8.20 at two of the three sites (Hi.0 and H2.0) and ranged from 50.2% to 63.7% of the efficiency of ABE8e at the third site (H3.0). Despite similarities in on-target editing, significant off-target editing was detected at all five off-target loci for ABE8e (ranging from 0.08 ± 0.02% to 4.64 ± 0.58%), while for all four ME-ABEs, off-target editing was only statistically significantly above background levels at the Hi.0 off-target 1 site (on-target editing was within error for all editors at this site). This activity ranged from 1.21 ± 0.28% for ABE7.10-HK to 1.81 ± 0.84% for ABE7.10-HSK, which was higher than that of ABE7.10 (0.19 ± 0.04%) but lower than that of ABE8.20 (3.18 ± 0.94%) and ABE8e (4.64 ± 0.58%). Reduced off-target activity at the Hi.0 site and negligible off-target activity at all other tested sites suggest that the improved on-target activity of the ME-ABEs is not coupled with an equivalent increase in their gRNA-dependent off-target editing profiles as is the case with ABE8e and ABE8.20.
To further evaluate gRNA-independent off-target activity, we used a previously described orthogonal R-loop assay (Supplementary Fig. 27a and Supplementary Discussion 11)13,41. Average on-target activity at two independent sites is plotted against average off-target activity at three independent, previously validated sites in Fig. 4d. A breakdown of these data by an on-target–off-target site combination can be found in Supplementary Fig. 27b–g. Across all three off-targets, the ME-ABEs showed similar levels of off-target editing activity to one another and to that of the parent ABE7.10, with substantially less off-target activity than ABE8e or ABE8.20 (Supplementary Fig. 28a). Averaged across all off-targets, editing efficiencies were 1.17 ± 0.14% for ABE7.10, 1.00 ± 0.09% for ABE7.10-HK, 0.91 ± 0.07% for ABE7.10-HRHSK, 10.15 ± 2.11% for ABE8e and 5.03 ± 0.22% for ABE8.20. The maximally reverted ABE7.10-HRHSK editor showed 11.2-fold and 5.6-fold reductions in average off-target editing activity compared with ABE8e and ABE8.20, respectively. For both on-target sites, the ME-ABEs facilitated higher editing efficiencies than ABE7.10 (Supplementary Figs. 28b,c and 29a). As expected, off-target editing efficiencies were independent of the on-target gRNA used (Supplementary Fig. 29b–d). Overall, the orthogonal R-loop data suggest that the ME-ABEs increased on-target editing activity independently of gRNA-independent off-target editing.
Finally, we assessed RNA off-target activity by performing whole-transcriptome RNA sequencing (RNA-seq) on the benchmark editors ABE7.10, ABE8e, ABE8.20 and the maximally modified ME-ABE ABE7.10-HRHSK. We found that all editors produced similar levels of A to I RNA off-target editing at levels significantly above that of the nCas9 control (Supplementary Fig. 30 and Supplementary Discussion 12). Taken together, these off-target analyses suggest that while the ME-ABEs reduce gRNA-dependent and gRNA-independent off-target DNA activity, they maintain RNA off-target editing levels at rates comparable to currently widely used editors.
ME-ABE reversions are ineffectual in ABE8e and ABE8.20 backgrounds, but offer advantages over other reported precision editors
Given the beneficial activity of ME-ABEs, we investigated combining the minimal and maximal sets of residue reversions by installing them into ABE8e and ABE8.20 (Extended Data Fig. 6). We found that applying these reversion combinations to the ABE8e and ABE8.20 backgrounds neither improved activity nor altered the editing window width, suggesting that the identified reversions’ activity was specific to the context of ABE7.10. Subsequent evolutionary changes in ABE8e and ABE8.20 may epistatically interfere with or override the beneficial interactions provided by ME-ABE reversions (Extended Data Fig. 6, Supplementary Figs. 20 and 31–35, and Supplementary Discussion 12). With this finding, we sought to further compare ME-ABEs with several additional editors engineered for precision (Fig. 5a). The ABE8e-V106W variant is reported to greatly reduce DNA and RNA off-target editing while maintaining high on-target activity, making it a favored editor for clinical applications7,13. To our knowledge, extensive editing window studies have not been published for this variant, although analogous TadCBE-V106W variants have been shown to have narrowed editing windows relative to their parent TadCBEs34. While the V106W variant was co-evolved alongside ABE8e, the ABE9 editor was rationally engineered from ABE8e for precision editing37. ABE9 contains two additional mutations (N108Q and L145T) and is reported to have an extremely narrow editing window centered at position 5 in the protospacer, alongside reduced off-target activity. To facilitate a direct comparison, these editors and maximally reverted editor ABE7.10-HRHSK were transfected into HEK293T cells with one of 12 gRNAs targeting a diverse set of endogenous genomic sites (Supplementary Fig. 18), and data were treated as previously described (Supplementary Discussion 8) to generate a representative editing window for all three editors (Fig. 5b–g and Supplementary Fig. 36). Using our defined metric for editing window, we found ABE8e-V106W to have an editing window spanning positions 3–11, matching that of its parent editor ABE8e. By contrast, ABE9 showed a narrower window encompassing only positions 5–6. When comparing average editing efficiencies at position 5, ABE7.10-HRHSK showed a 1.6-fold increase in editing over ABE9, while the ABE8e-V106W variant showed an additional 1.1-fold increase over ABE7.10-HRHSK.
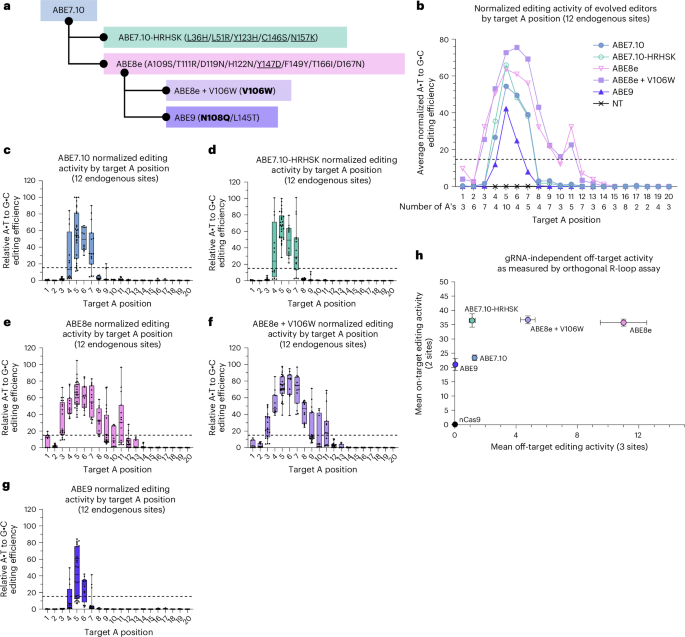
a, The subset of editors analyzed for precision editing are shown, highlighting derivation ancestry. Residues that differentiate each precision editor from their parent editor are listed—those that correspond to the wtTadA identity (reversions) are underlined, those that have been modified multiple times over the course of engineering (‘evolutionary hot spots’) are shown in bold, and all others are in plain text. b, HEK293T cells were transfected with plasmids encoding one of the listed ABEs and one of 12 gRNAs targeting an endogenous site or an NT control (Supplementary Fig. 18; indicated by asterisks). After a 72-h incubation, genomic DNA was collected, target sites were amplified and sequences were analyzed by NGS. Editing was performed in n = 3 biological replicates and normalized as described in Fig. 4. Normalized editing scores for all 96 A’s were then sorted by A position, and scores were further averaged across gRNAs to identify the editing window. The number of A’s considered for each position is given below the x-axis. The horizontal dotted line shows the 15% cutoff used to determine the editing window for each editor. c–g, Data from b are shown for editors ABE7.10 (c), ABE7.10-HRHSK (d), ABE8e (e), ABE8e + V106W (f) and ABE9 (g). Points show normalized editing efficiency for a given editor at a given site and are shown for all replicates. Box hinges illustrate quartiles, lines show medians and bars show minimum and maximum editing. h, Orthogonal R-loop assay was used to analyze gRNA-independent off-target activity of precision editors as described in Fig. 4d. The x–y plot indicates the average off-target activity for the most edited A position across three independent off-target sites, and average on-target activity at the most edited A position across two independent on-target sites, accounting for n = 3 biological replicates with error bars showing propagated standard deviation.
Source data
We additionally evaluated the ABE7.10-HRHSK variant against ABE8e-V106W and ABE9 using the orthogonal R-loop assay to assess gRNA-independent off-target editing activity (Fig. 5g and Supplementary Figs. 37 and 38). Using the same on- and off-target editing sites as before, we found that ABE9 had nearly undetectable levels of gRNA-independent off-target editing activity, with an average off-target efficiency of 0.05 ± 0.02%. ABE8e-V106W showed an average off-target efficiency of 4.77 ± 0.48%, representing a 2.3-fold decrease from ABE8e. However, the average off-target efficiency of ABE7.10-HRHSK was reduced 4.2-fold compared with ABE8e-V106W. The average on-target editing activities at the two selected sites suggest that ABE7.10-HRHSK and ABE8e-V106W have similar on-target editing efficiencies (36.6 ± 2.4% and 36.8 ± 1.4%, respectively), while that of ABE9 (21.1 ± 2.0%) was 1.7-fold less than that of ABE7.10-HRHSK. These comparisons with other precision editors suggest the ME-ABEs strike a unique balance between precision and activity. While the ME-ABE editing window is larger than that of ABE9, on-target editing is greatly improved in comparison, and on-target activity is comparable to that of ABE8e-V106W but with reduced bystander and gRNA-independent off-target editing.
ME-ABEs facilitate modeling of challenging SNVs of clinical interest
Given our findings that the ME-ABEs generally increased editing activity relative to ABE7.10, improved editing at multiple sequence contexts, and on average matched the efficiency of ABE8e and ABE8.20 at the central A5 position without widening the editing window, we reasoned that these alternative tools could expand our ability to robustly and precisely edit target A’s beyond well-established sites in HEK293T cells. We found that the ABE7.10-HRHSK ME-ABE maintained similar editing profiles in different cell types (Fig. 6a,b, Extended Data Fig. 7 and Supplementary Discussion 14). In addition, ME-ABEs successfully edited six SNVs of clinical interest that had previously evaded modeling with base editors ABE7.10, ABE8e and ABE8.20 owing to the presence of missense-mutation-inducing bystanders, target A sequence context, overall low editing efficiencies or a combination of these issues (Fig. 6c–g and Supplementary Figs. 39–44). For a further discussion on site selection and data generation and normalization, see Supplementary Discussion 15.
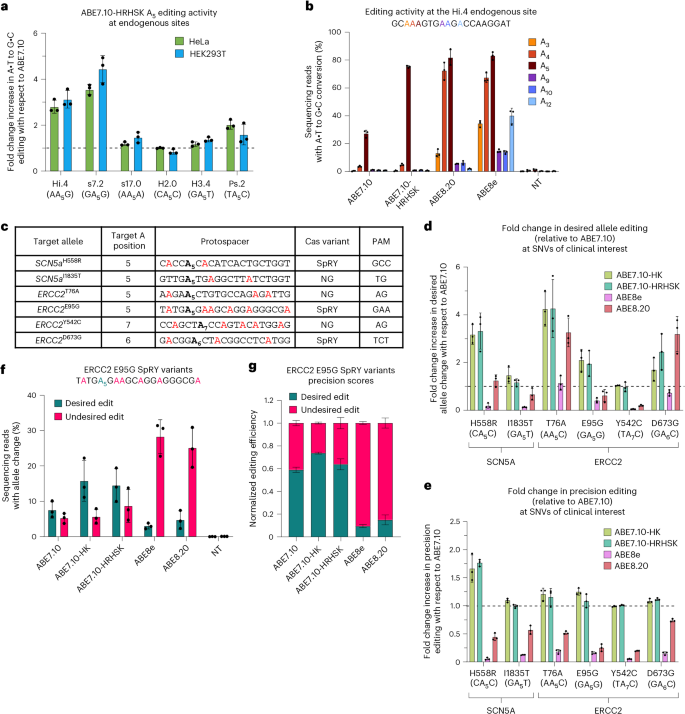
a, Activity of ABE7.10-HRHSK was measured at six genomic sites in both HEK293T and HeLa cells. HEK293T cells were treated as described in Fig. 4. HeLa cells were transfected with plasmids encoding ABE-P2A-mCherry and one of six gRNAs. The mCherry fluorescence marker enabled sorting for transfected cells before genomic DNA collection and NGS analysis. Activity is presented as an average fold change in activity relative to ABE7.10 for the indicated A; n = 3 biological replicates, and error bars show standard deviation. b,Editing activity of various ABEs at the Hi.4 site in HeLa cells. Bars show average editing of n = 3 biological replicates, and error bars represent the standard deviation. c, Six SNVs of clinical interest with challenging-to-edit target A’s were selected for modeling with ABEs. The target A is shown in bold, while bystander A’s whose edit would result in missense mutations are shown in red. d, Summary of editing activity at six SNVs of clinical interest. Data for each editor are presented as fold changes in the percentage of DNA sequencing reads with the desired allele sequence (that is, target A edited and no bystander editing) with respect to ABE7.10. Bars show mean fold change for n = 3 independent sets of transfections, error bars indicate standard deviation and the horizontal dashed line shows average ABE7.10 activity. e, Summary of editing precision at six SNVs of clinical interest. Precision scores for each editor were calculated by determining the percentage of all edited sequencing reads in which only target A was appropriately modified. Fold-change values were determined by taking the average precision score for each editor and comparing it with that of ABE7.10 for n = 3 biological replicates; error bars show standard deviation and fold-change values from individual replicates are shown as points. f, Absolute editing efficiencies of ABEs used to introduce the ERCC2E95G SNV. Absolute editing was measured in n = 3 biological replicates, shown as individual points; bars show means, and error is reported as standard deviation. g, The absolute editing efficiency data from f was normalized to total editing (all sequencing reads with any type of edit) to report precision of desired editing among all edited sequences. Bars show average precision scores for n = 3 biological replicates, and error bars represent standard deviation
Source data
At five of the six tested sites, both ME-ABEs showed higher absolute editing efficiencies than ABE7.10 (Fig. 6d), ranging from a 1.17 ± 0.15-fold higher editing rate for ABE7.10-HRHSK at the SCN5AI1835T site to a 4.26 ± 1.21-fold higher editing rate for the ABE7.10-HRHSK editor at the ERCC2T76A site. The ERCC2Y542C edit was the only site tested where activity of the ME-ABEs matched that of ABE7.10 without further improvement. However, we will note that the target A for this site lies at position 7 rather than 5. These results therefore are consistent with our characterization studies (Fig. 4b), which showed that the ME-ABEs have enhanced editing relative to ABE7.10 at positions 4–6, while maintaining similar editing rates at the remaining editable positions.
At all sites, both ME-ABEs showed vastly improved precision over ABE8e (Fig. 6e). This ranged from a 6.6- and 6.3-fold increase in precision over ABE8e for ABE7.10-HK and ABE7.10-HRHSK, respectively, for the ERCC2T76A mutation, to a 28.9- and 30.2-fold increase over ABE8e for ABE7.10-HK and ABE7.10-HRHSK, respectively, for the SCN5AH558R mutation. Similarly, although to a lesser degree, the ME-ABEs improved precision over that of ABE8.20 at all sites, ranging from 1.5-fold increases for both ABE7.10-HK and ABE7.10-HRHSK at the ERCC2D673G mutation to a 5.0- and 5.1-fold increase for ABE7.10-HK and ABE7.10-HRHSK, respectively, at the ERCC2Y542C mutation. While the ABE8 variants were often more ‘active’ than the ME-ABEs, resulting in a greater number of edited sequencing reads, their tendency to edit multiple A’s present in their wider editing windows led to less precise editing outcomes. This was particularly well illustrated at the ERCC2E95G site (Fig. 6f,g and Supplementary Fig. 42). While the target A5 was readily edited by ABE8e and ABE8.20 (with overall bulk editing efficiencies of the target A of 34.0 ± 4.8% and 32.1.0 ± 7.2%, respectively), adjacent bystanders including a readily edited A7 were also modified in many sequences, resulting in high levels of undesired editing efficiency and low precision scores (9.5 ± 1.28% and 15.1 ± 4.4% of edited reads had only the target A•T, and no bystanders, converted to G•C for ABE8e and ABE8.20, respectively). While bulk editing efficiency of the target A by ABE7.10-HRHSK at this site was 23.8 ± 7.5%, which is lower than that of ABE8e and ABE8.20, 63.8 ± 4.8% of edited reads had only the target A, and no bystanders, edited, a 6.7- and 4.2-fold increase in precision editing compared with ABE8e and ABE8.20, respectively. At all six sites tested, the ME-ABEs had precision scores on par or greater than ABE7.10, and always greater than ABE8e and ABE8.20. Furthermore, at all but one site, ME-ABEs showed higher precise allele editing efficiencies than ABE8 editors. Overall, we present use of the ME-ABEs in different Cas architecture contexts and show the utility and advantages of editors with improved editing efficiency but narrow editing windows for modeling SNVs of clinical interest.


