Antibiotic Resistance is Ancient Feature of Microbial Life, New Review Says

The spread of antibiotic resistance genes poses a serious and global threat to public health. A comprehensive new study by scientists at Hohai University explores the evolutionary origins, ecological factors underlying the proliferation and spread of antibiotic resistance genes, and their far-reaching environmental implications.
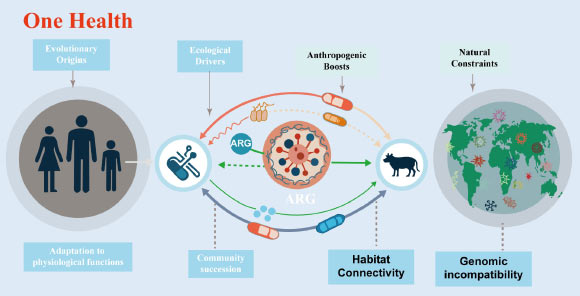
The evolution of antibiotic resistance genes is linked to intrinsic physiological roles and ecological divisions. Image credit: Xu and others., doi: 10.48130/biocontam-0025-0014.
Antibiotic resistance genes have emerged as one of the most pressing global threats to public health, with their dissemination across the interconnected compartments of humans, animals, and the environment.
They have been detected in some of the most extreme and pristine environments on the planet, including the depths of the Mariana Trench, the pristine soil of Alaska, and 30,000-year-old permafrost sediments, environments completely untouched by anthropogenic exposure to antibiotics.
This wide distribution confirms an essential truth: these bacterial species evolved the ability to tolerate antibiotics millions of years before the production of antibiotics for clinical and agricultural applications.
“Antibiotic resistance did not start with modern medicine,” said Dr. Guoxiang You, corresponding author of the study.
“Many resistance genes originally evolved to help bacteria survive environmental stresses, long before humans discovered antibiotics. »
“The real danger today comes from the way human activities break down natural barriers and allow these genes to spread into pathogens. »
“Many resistance genes derive from ordinary bacterial genes with essential physiological roles, such as pumping out toxic substances or transporting nutrients,” the researchers explained.
“Through evolution, these genes acquired the ability to defend against antibiotics as a secondary function.”
In undisturbed ecosystems such as soils, lakes, and remote environments, most resistance genes remain locked into specific microbial communities and pose little risk to human health.
“One of the main reasons for this containment is genomic incompatibility,” they added.
“Bacteria that are genetically very different often cannot easily exchange and use resistance genes. »
“This natural mismatch acts as a biological firewall, limiting the spread of resistance between species and habitats.”
“However, human activity is weakening this firewall.”
In this review, the authors highlight how agriculture, wastewater discharges, urbanization and global trade are increasing connectivity between once-separate environments.
Antibiotics used in medicine and animal agriculture create strong selection pressures, while manure spreading, wastewater reuse, and environmental pollution bring together bacteria from soil, animals, and humans.
These conditions facilitate the penetration of resistance genes into pathogenic microbes.
“Human-induced habitat connectivity changes everything,” said Dr Yi Xu, first author of the study.
“When bacteria from different environments are repeatedly brought into contact under antibiotic pressure, once-harmless resistance genes can become a serious threat to public health.”
“Wastewater treatment plants are identified as critical hotspots, where high bacterial densities and residual antibiotics promote genetic exchange. »
“Agricultural soils fertilized with manure can also serve as bridges, allowing resistance genes to pass from livestock to environmental bacteria and eventually back to humans through food, water or direct contact.”
Importantly, scientists emphasize that not all resistance genes are equally dangerous.
High abundance in the environment does not automatically mean high risk.
Understanding which genes are mobile, compatible with human pathogens, and linked to disease is essential for effective surveillance and control.
Researchers call for ecosystem-focused strategies to combat antibiotic resistance.
These include reducing unnecessary antibiotic use, improving wastewater treatment technologies, more carefully managing manure and sludge, and protecting relatively pristine ecosystems that serve as a benchmark for natural resistance levels.
“Antibiotic resistance is not just a medical problem,” Dr. You said.
“It’s an ecological problem rooted in the way we interact with the environment.”
“Protecting antibiotics for future generations requires protecting the integrity of ecosystems today. »
“By integrating evolutionary biology, microbial ecology and environmental sciences, a One Health approach offers the most realistic path to tackling one of the greatest global health challenges of our time. »
The review was published online on December 5, 2025 in the journal Biocontaminant.
_____
Yi Xu and others. 2025. Evolutionary origins, ecological factors and environmental implications of the proliferation and dissemination of antibiotic resistance genes: a “One Health” perspective. Biocontaminant 1:e014; doi: 10.48130/biocontam-0025-0014



