Physicists create electron ‘catapult’ that moves particles through solar cells at record speed

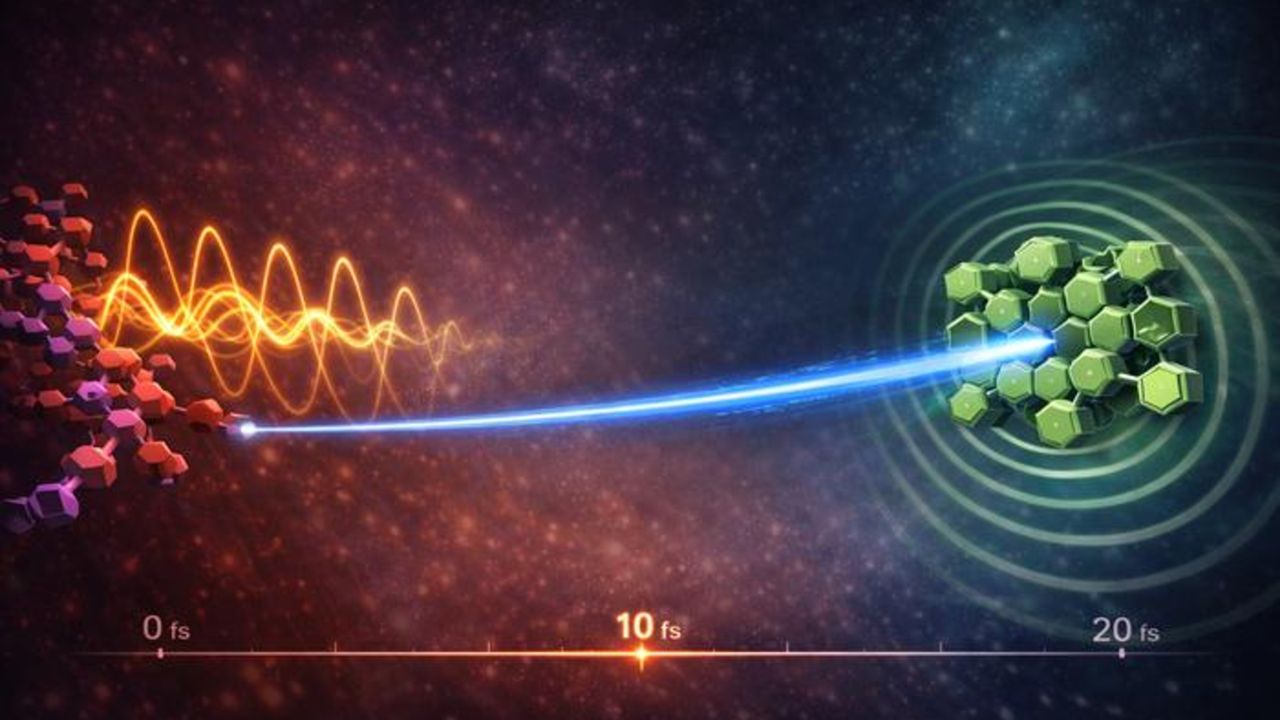
Molecular vibrations can “catapult” electrons through solar materials in quadrillions of a second – much faster than previously thought, according to a new study.
The findings could help scientists find more efficient ways to convert solar energy into electricity, according to the study published March 5 in the journal Natural communications.
Article continues below
Organic molecules become solar
Organic solar cells use carbon-based molecules, rather than silicon, to convert sunlight into electricity. In theory, organic solar cells could provide this electricity more cheaply than conventional solar cells, but they are much less efficient.
In a typical organic solar cell, an electron donor and an electron acceptor are sandwiched between two conducting electrodes. When light hits the cell, it generates a “excitement“, an electron-hole pair. Excitons split at the interface between the donor and acceptor, generating electricity.
Seeing this happen on this time scale within a single molecular vibration is extraordinary.
Pratyush Ghosh, researcher at the University of Cambridge
To achieve rapid charge transfer at the interface and limit energy loss, donor and acceptor molecules generally have strong electronic coupling, or overlap between their electronic states, which allows charges to move easily between molecules. They also often have a large energy difference between them, but this limits the voltage available from the device.
In the new study, researchers observed ultrafast charge transfer at the junction between the electron donor and electron acceptor in an organic solar cell, without needing to conform to any of these constraints. The team used a short laser pulse to excite the electron donor, a polymer called TS-P3, then used another laser to measure how the system changed during charge transfer.
This charge transfer happened in 18 femtoseconds, about as fast as an individual molecule vibrates. A few other systems without large driving forces exhibit charge transfer in excess of 100 to 200 femtoseconds, but most take ten to a thousand times longer.
“To see this happen on this time scale within a single molecular vibration is extraordinary,” Ghosh said in the release.
A “molecular catapult”
This similar timing was no coincidence. In a second set of laser experiments, the team found that vibrations in the polymer’s donor molecule launched an electron across the junction to an acceptor molecule. When the electron arrived, it triggered superimposed vibrations in the acceptor molecule. This overlap allowed the charge transfer to occur much faster than expected, and without requiring strong coupling or a large energy difference.
“Instead of drifting randomly, the electron is launched in a single coherent burst,” Ghosh said in the release. “The vibration acts like a molecular catapult. The vibrations not only accompany the process, they actively drive it.”
The results help explain the processes that control the speed of charge transfer and establish new strategies for designing more efficient organic solar cells and materials, the researchers wrote in the study.
“Instead of trying to eliminate molecular motion, we can now design materials that use it, transforming vibrations from a limitation into a tool,” study co-author Akshay Raophysicist at Cambridge, said in the release.
Ghosh, P., Royakkers, J., Londi, G., Giannini, S., Arul, R., Gillett, AJ, Keene, ST, Zelewski, SJ, Beljonne, D., Bronstein, H. and Rao, A. (2026). Vibration-assisted subcycle charge transfer to a non-fullerene acceptor heterojunction. Natural communications, 17(1). https://doi.org/10.1038/s41467-026-70292-8




