Physicists found a novel way to peer inside a radioactive molecule — and revealed a strange phenomenon in its nucleus

Physicists have studied a rare molecule for the first time to observe how magnetism is distributed in a radioactive nucleus.
The rules of nature generally do not change. If you throw a ball in Seattle or Tokyo, it falls the same way. Physicists call this “symmetry,” and they use symmetry as a guide to how the universe should behave. This is what keeps the world coherent: if the laws of physics worked differently on Tuesday, the universe would be chaos.
But some parts of nature don’t seem to respect this perfect balance. For example, it may seem fair to assume that the universe should treat matter and antimatter equally. Yet our universe is made almost entirely of matterand physicists still don’t know why.
A promising place to look for answers is inside radioactive nuclei. Indeed, the unequal arrangement of protons and neutrons can amplify the smallest symmetry breaks. If scientists can detect these small asymmetries, it could reveal new physics beyond the Standard Model, according to Silviu-Marian Udrescuphysicist at MIT and co-author of a new study on the phenomenon.
In a study published on October 23 in the journal ScienceScientists from CERN and MIT examined a short-lived radioactive molecule called radium monofluoride (RaF) to measure its energy spectrum. But, surprisingly, they ended up making the first observation of how magnetism is distributed in one of its cores. This phenomenon, known as the Bohr-Weisskopf effect, has never been observed in a molecule before.
The atom’s lawyer
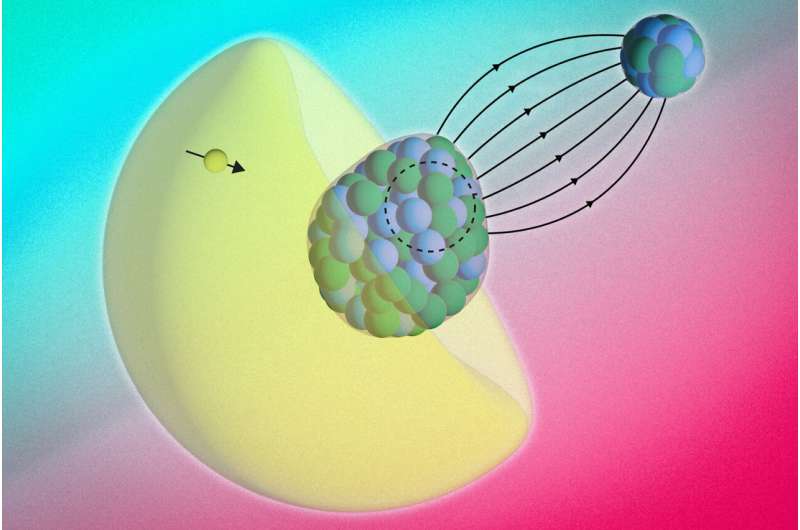
The RaF molecule is made up of two atoms: radium and fluoride. Each with its own core. The radium nucleus has a property called octupole deformation.
“You can think of it as the kernel itself being shaped like a pear or an avocado,” Shane Wilkinsphysicist at MIT and first author of the study, told Live Science. Due to its asymmetric shape, RaF is an ideal candidate for finding the asymmetries the team is looking for.
“It’s a very rare property,” Udrescu added. “This only happens in a few handfuls of atomic nuclei across the entire nuclear diagram. And all of those nuclei that have this pear shape are radioactive.”
This radioactivity makes these nuclei difficult to study because these isotopes are unstable and short-lived. This means they disintegrate in about 15 days and can disappear before researchers can make many measurements. “We can only produce them in very small quantities,” Wilkins said.
The Bohr-Weisskopf effect has been observed in individual atoms, where electrons interact with a single nucleus. However, its detection inside a molecule is more difficult. This is because electrons are constantly moving between the two nuclei. Movement can scramble magnetic signals and make them harder to detect. In a RaF molecule, the fluoride atom is a simpler binding partner. This allows scientists to focus on the magnetic structure of the heavier radium nucleus.
The team created radium monofluoride for the first time CERN ISOLDE facility. They exploded a uranium target with high-energy protons to produce the rare isotope radium-225 and combined it with fluorine gas. Each molecule only existed for a fraction of a second. The researchers were only able to detect around fifty per second in the right state for measurement.
Next, they aimed several laser beams of slightly different frequencies at the molecules. When the molecule absorbed or emitted light, the scientists recorded the tiny changes in that light. This produced a spectrum. Normally, these models tell scientists how electrons move around the nucleus. But in this case, some changes revealed that the electrons were influenced from inside the nucleus.
“The electron actually probes inside the core, so you can no longer treat it as a long-range interaction. Instead, it begins to detect the internal properties of the radium core itself,” Wilkins said.
“This effect is called the Bohr-Weisskopf effect,” Wilkins added. “To the best of our knowledge, this has never been seen in a molecule before. The fact that we can both observe this effect experimentally and describe it theoretically tells us a lot about the suitability of these molecules for future precision measurements.
Now that researchers have mapped the internal structure of RaF, they can use it to probe even smaller effects that could break nature’s symmetries. The next step, Wilkins said, is to slow down and trap these molecules with lasers to make even more precise measurements.
“We now know that they can be powerful tools for searching for new physical data,” Udrescu said.



