Sleeping sickness could be wiped out with this new drug : NPR

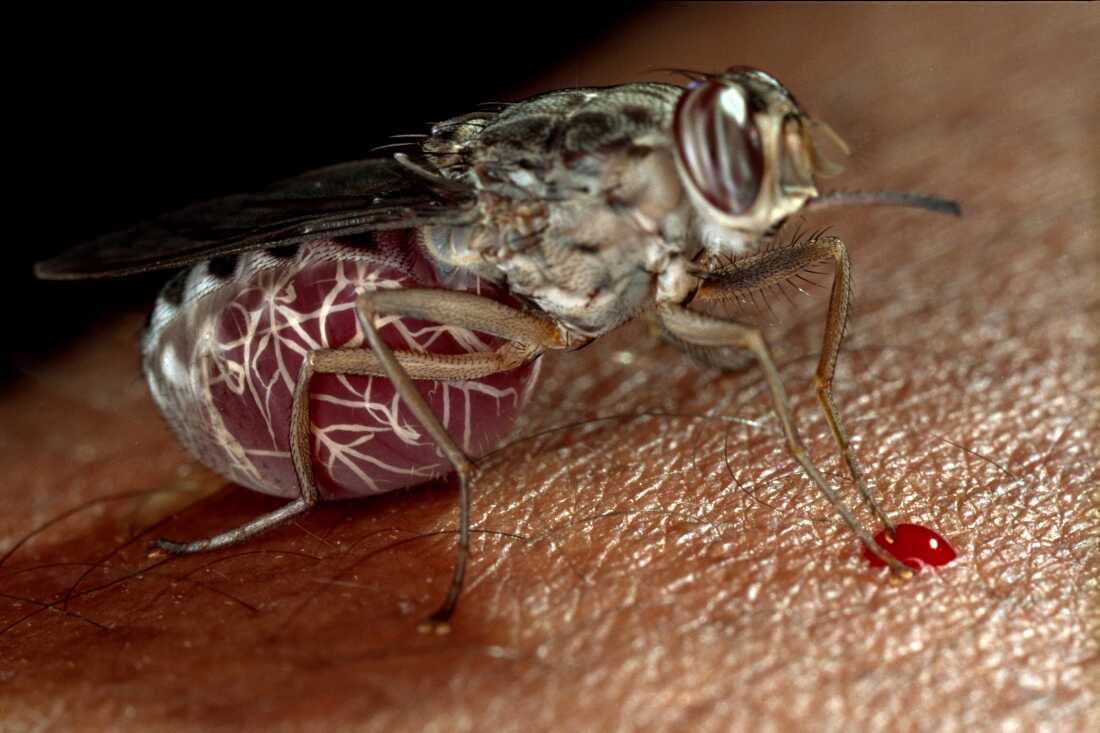
A tsetse fly: its bite can spread the parasite responsible for sleeping sickness.
Patrick Robert/Corbis/Sygma/via Getty Images
hide caption
toggle caption
Patrick Robert/Corbis/Sygma/via Getty Images
Sleeping sickness is a notorious illness — immortalized in Joseph Conrad’s book Heart of Darkness. It only takes one bite from a tsetse fly carrying the parasite to infect someone. Without treatment, one form of the disease can progress from mild symptoms to death within a few weeks.
Now a new drug promises to help the World Health Organization reach its goal of eliminate the disease by 2030. A European Medicines Agency committee has given the green light to the first single-dose treatment, a drug called acoziborole, which could be used early next year.
Acoziborole is particularly notable because it is taken as three tablets swallowed together in a single dose, replacing long-used previous treatments that included intravenous drugs known to cause a “burning” sensation in the veins and be fatal for nearly one in 20 patients. Even the current first-line oral treatment, fexinidazole, must be taken for 10 days and comes with serious side effects such as nausea, vomiting and heart rhythm disturbances. In contrast, clinical trials of acoziborole found only one significant side effect: mild to moderate headache.
“For decades, available treatments were difficult to use,” says Dr. Gerardo Priotto, who leads the World Health Organization’s efforts against sleeping sickness and was not part of the team developing the new drug. Therapies required staff, equipment and reliable infrastructure, he said. “These challenges were particularly severe in isolated rural areas, where most cases occur and health services are limited.”
“A tool for transformation”
The new drug, acoziborole, eliminates almost all of these barriers, representing a major breakthrough. “Its well-tolerated single-dose regimen can significantly simplify patient care, improve access to treatment, and accelerate progress toward eliminating sleeping sickness.” This is a transformative tool for patients and public health programs,” says Priotto.
Severe side effects of previous drugs also discouraged people from seeking treatment, says Dr. Stéphane Hugonnet, who worked on clinical trials and leads the sleeping sickness response for the Drugs for Neglected Diseases Initiative (DNDi), a nonprofit that developed the drug with pharmaceutical company Sanofi and with funding from several partners, including the Gates Foundation, which financially supports NPR’s global health coverage.
“Many patients remember what treatment was like for themselves or their family members and were afraid of the care,” says Hugonnet.
The tsetse fly picks up the parasite from an infected person and can then pass it on to someone else. The insect thrives in warm wooded savannahs and in vegetation along lakes or streams, so the disease tends to take hold in remote areas where people rely on fishing, hunting and agriculture for their livelihood.
“Like many tropical diseases, sleeping sickness is a disease of the poor,” says Dr. Peter Hotez, dean of the National School of Tropical Medicine at Baylor College of Medicine in Houston.
Over the years, hundreds of thousands of people have died from sleeping sickness, but sustained efforts – tsetse fly control, diagnostic testing and medical treatment – have significantly reduced the incidence. The current annual number of cases is around 1,000 worldwide, almost two-thirds of them in the Democratic Republic of Congo. But “sleeping sickness has ebbed and flowed at many points in history,” says Hotez.
Sleeping sickness caused by Trypanosoma brucei gambiense the parasite accounts for more than 90% of cases, and it is the target of the new drug. The disease progresses in two phases: an early, less severe phase marked by fever and headache, then, once the parasite crosses the blood-brain barrier, a later phase with more serious neurological effects, such as confusion and seizures. At this point, the parasite disrupts regions of the brain that regulate the circadian rhythm, causing patients to become sleepy during the day and awake at night. Without treatment, the disease can progress to coma and death.
Some previous treatments have only worked for one stage of the disease, but acoziborole treats both.
“We can’t ask for anything better than this,” says Dr Wilfried Mutombo Kalonjihead of clinical operations in West and Central Africa for DNDi, which began its work in 2003 after recognizing that many diseases in low-income countries were curable, but that pharmaceutical companies had little financial incentive to develop treatments for people and countries unable to pay for them.
Try to speed up the treatment
Kalonji spoke to NPR from Sud-Ubangi, a remote province in the Democratic Republic of Congo, where he helped lead clinical trials submitted to the European Medicines Agency. He is currently overseeing a new trial to determine whether blood (serological) tests could allow treatment to be started immediately rather than waiting for confirmatory tests, which take longer, cost more and generally cannot be done in the field.
Monica Mungier, who studies sleeping sickness at the Johns Hopkins Bloomberg School of Public Health but was not involved in developing the new drug, says the parasite can sometimes escape detection. A negative test therefore does not always mean that a patient is not infected.
However, if it is determined that the blood tests are reliable enough to guide care, “then testing and treatment can begin the same day,” Kalonji says. Such a change could increase the number of patients treated and potentially end disease transmission because human reservoirs would disappear, Mungier says.
The next step is a review of acoziborole by the Democratic Republic of Congo’s Ministry of Health and the WHO to see whether the agency should update its treatment guidelines, which would help other countries authorize use of the drug. But global health experts fear that future cuts in U.S. and Western funding could prevent the drug from reaching countries that need it.
Much of the progress toward eliminating sleeping sickness rests on the work of African researchers and patients who participated in the trials, despite difficult conditions. “Clinical trials are difficult,” Kalonji says. “Patients are often in remote areas without electricity or water. We had to put all of this in place, including training health workers, providing internet connections, electricity and a safe way to access the testing sites since they were remote. We overcame the challenges.”
Fran Kritz is a health policy reporter based in Washington, D.C. and a regular contributor to NPR. She also reports for The Washington Post And Very good health.




