Lyme disease is spreading, but a new vaccine could curb infections

April 9, 2026
4 min reading
Add us on GoogleAdd SciAm
Lyme disease is spreading: a new vaccine could curb infections
A new Lyme disease vaccine made by Pfizer and Valneva could reduce infection rates from the tick-borne illness, but federal approval and patient adoption could pose a challenge.
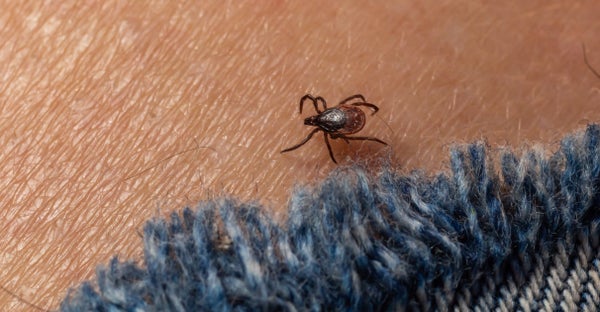
Oleg Marchak via Getty Images
Since pharmaceutical companies Pfizer and Valneva announced positive results from their Phase 3 trial of a vaccine to prevent Lyme disease last month, two questions have arisen: Will the vaccine receive approval from the Food and Drug Administration? And if so, will the American public accept it?
An effective vaccine could significantly reduce the growing burden of Lyme disease. Populations of ticks that spread the disease are increasing, and nearly half a million cases of Lyme disease occur each year in the United States. But the vaccine must overcome the same hurdles that led to the withdrawal of a previous Lyme vaccine from the market in 2002.
The recent announcement showed that the vaccine was able to prevent Lyme disease infection. But its effectiveness was only about 73 to 75 percent. The trial also suffered several setbacks, including quality issues that led Pfizer to drop about half of the study population several years ago. The results were also not as statistically powerful because fewer infections occurred than expected in the placebo group during the trial. Still, clinicians approach the latest news with cautious optimism. “I think it’s great news,” says Martin Backer, an infectious disease physician at NYU Langone Health who participated in a previous pediatric trial of this vaccine. “Time will tell if it’s acceptable.”
On supporting science journalism
If you enjoy this article, please consider supporting our award-winning journalism by subscription. By purchasing a subscription, you are helping to ensure the future of impactful stories about the discoveries and ideas shaping our world today.
Lyme disease is caused by Borrelia burgdorferi bacteria carried by Scapular ixodes, the blacklegged tick, or deer tick. The tick transmits the bacteria to humans when it attaches to human skin and feeds on blood. Although the disease is mild in some people, if left untreated it can cause serious neurological, heart and joint inflammation problems and lasting symptoms in others, says Daniel Pastula, a neurologist at the University of Colorado Anschutz. Prompt treatment with antibiotics can prevent this damage, but cases are often missed, testing is complex, and the disease is often underdiagnosed.
More than 89,000 cases of Lyme disease were reported to the Centers for Disease Control and Prevention in 2023, but the agency estimates the country’s true burden at about 476,000 cases per year. Cases have increased, the tick’s geographic distribution has expanded, and tick season is starting earlier and lasting longer, in part because of climate change.
Using insect repellent, wearing protective clothing such as long pants and long sleeves, and checking for ticks after being outside in tick-prone areas can significantly reduce the risk of a bite. But most people don’t follow these strategies, says Pastula.
The new vaccine is what is called a protein subunit vaccine. Called LB6V, it targets six serotypes, or bacterial variations, of the skin’s outer surface protein A (OspA). B. burgdorferi bacteria responsible for Lyme disease. This triggers the immune system to develop antibodies against the six most common subtypes of the bacteria in North America and Europe. Unlike most vaccines, which target pathogens present in the body, this one targets the bacteria present in the tick. When a tick feeds on the blood of a vaccinated person, it ingests these antibodies, which then bind to the bacteria in the tick and prevent them from entering the person.
The vaccine first showed effectiveness in mice in 2017, leading the FDA to grant it fast-track designation, and the phase 1 trial showed no major safety concerns in humans. The recent Phase 3 trial involved approximately 9,400 participants ages five and older in the United States, Canada and Europe. Participants received three doses of the vaccine over a period of five to nine months, then a fourth booster dose before the start of a second tick season.
The new vaccine has some similarities to LYMErix, a vaccine approved by the FDA in 1998 that was later withdrawn due to public concerns about safety (some vaccine recipients reported musculoskeletal pain that they attributed to the vaccine) and poor market performance. In their respective trials, both vaccines had less than 80 percent effectiveness and uncertain duration of immunity. Notably, people will likely need booster shots before each tick season, says Ondrej Hajdusek, a parasite biologist at the Czech Academy of Sciences, who co-authored a paper on the phase 1 trial of the new vaccine in the United States. Lancet. Antibodies often disappear over time, so a booster would ensure there are enough antibodies present for ticks to ingest each season.
Despite its similarities to the previous vaccine, LB6V “reflects lessons learned from the LYMErix era,” says Hajdusek. The new vaccine has been tested in children under 15 years old. It covers more serotypes of the bacteria and does not contain the component of the LYMErix vaccine thought to cause arthritic pain. The proposed link between the earlier vaccine and arthritis was never confirmed by subsequent safety research, but fear of an association was among the factors that caused the vaccine to be withdrawn from the market.
Lorraine Johnson, CEO of LymeDisease.org, a nonprofit education and advocacy organization, says she hoped to see a vaccine that would protect against the various co-infections that can occur with Lyme disease. In the meantime, she wants to see safety data when companies release this information.
“Lyme patients want a safe and effective vaccine,” says Johnson, “and they want [the manufacturer to be] truly honest about disclosing adverse events.
A Pfizer spokesperson said Scientific American that the company is not giving interviews about the vaccine at this time.
Pfizer has submitted LB6V vaccine trial data to the FDA. If the vaccine is approved, the CDC’s Advisory Committee on Immunization Practices (ACIP) would normally determine who to recommend it to. (A recent court ruling had blocked previous appointments of current ACIP members.) The recommendation would likely involve assessing who is at risk for exposure to Lyme disease, similar to recommendations for travel vaccines, Backer says.
At this point, says Hajdusek, “the main practical question will probably be public acceptance, particularly if an annual pre-season booster is recommended.”
It’s time to defend science
If you enjoyed this article, I would like to ask for your support. Scientific American has been defending science and industry for 180 years, and we are currently experiencing perhaps the most critical moment in these two centuries of history.
I was a Scientific American subscriber since the age of 12, and it helped shape the way I see the world. SciAm always educates and delights me, and inspires a sense of respect for our vast and beautiful universe. I hope this is the case for you too.
If you subscribe to Scientific Americanyou help ensure our coverage centers on meaningful research and discoveries; that we have the resources to account for decisions that threaten laboratories across the United States; and that we support budding and working scientists at a time when the value of science itself too often goes unrecognized.
In exchange, you receive essential information, captivating podcasts, brilliant infographics, newsletters not to be missed, unmissable videos, stimulating games and the best writings and reports from the scientific world. You can even offer a subscription to someone.
There has never been a more important time for us to stand up and show why science matters. I hope you will support us in this mission.




