Cancer vaccine shows promise against HPV-related throat tumors in early study

A vaccine designed to fight head and neck cancers caused by HPV has shown promising results in a laboratory study of human tissue and mice.
If proven effective in humans, the therapeutic vaccine could complement standard cancer therapies, and its design could help scientists develop better vaccines against other diseases.
The vaccine Gardasil 9 can prevent HPV infections and thus reduce the risk of these cancers in the long term. But for people who already have HPV-related tumors, treatment still relies on surgery, radiotherapy and chemotherapy. Combining a cancer vaccine with these conventional therapies could improve their effectiveness by teaching the immune system to fight cancer.
Today, scientists have developed a cancer vaccine whose components are arranged in a unique structure. Similar to preventative vaccines, cancer vaccines train the immune system to recognize specific proteins – in this case, a protein found on HPV-positive tumors – and often contain ingredients called adjuvants that stimulate the immune response. Rather than preventing the disease in the first place, cancer vaccines are typically used to treat the disease and help prevent its recurrence.
In laboratory studies of HPV-positive head and neck cancer, this carefully designed new vaccine slowed tumor growth and improved survival in mice, according to a study published Wednesday (Feb. 11) in the journal Scientific advances.
Dr. Ezra Cohenhead and neck cancer specialist at UC San Diego Health who was not involved in the study, said that if the vaccine works in humans, it could complement standard therapies.
“We can imagine a multimodal approach to make a patient disease-free, then a vaccine to prevent recurrences,” he said. But he cautioned that results obtained with laboratory animals and isolated tissues do not always translate to results in humans. “The real test is in people,” he told Live Science in an email. “But strong preclinical data, like this, increases the chances of clinical trial success.”
In this case, the underlying design of the vaccine is remarkable.
“The main conclusion is that the structure of the vaccine makes a significant difference,” Cohen said. “Successful vaccination is not just about selecting the right antigens [target proteins] but by placing these antigens in the correct order with other elements of the vaccine. »
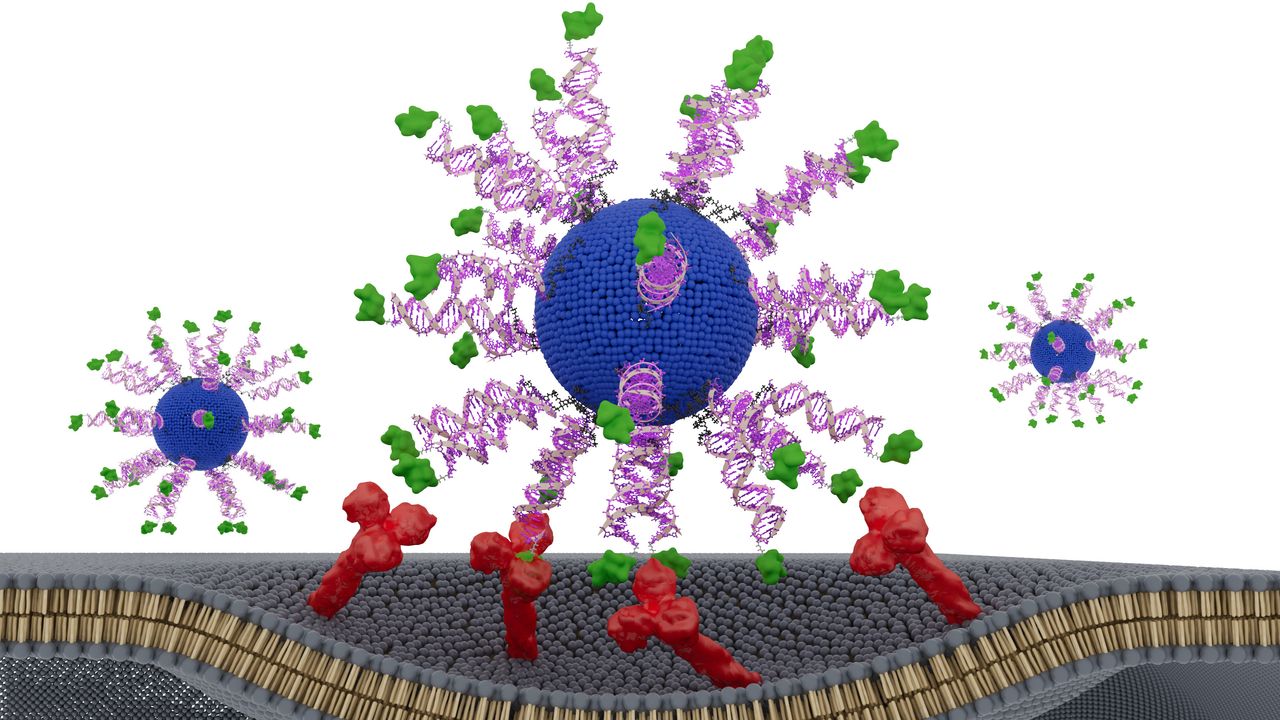
The vaccine uses spherical nucleic acids (SNA) – globe-shaped DNA particles that enter immune cells and bind to targets. more efficiently than linear DNA do. Each SNA nanoparticle in the vaccine consists of a fatty core surrounded by an adjuvant and a fragment of an HPV protein from tumor cells. The adjuvant mimics bacterial DNA and is recognized by the immune system as “foreign”.
The researchers tested three models, changing only the way the HPV fragment was positioned. One version hid it inside the nanoparticle, while the other two versions had the HPV fragment on the surface of the particle, attached to different ends of the fragment structure, known as the N terminus and the C terminus.
The version with the fragment attached to the surface via its N terminus triggered the strongest immune response, the team found. This design led killer T cells – immune cells that destroy infected, damaged and cancerous cells – to produce up to eight times more interferon gamma, a key anti-tumor signaling protein. This made them more effective at killing HPV-positive cancer cells.
In mouse models of HPV-positive cancer, the vaccine significantly slowed tumor growth. Additionally, when tested on tumor samples taken from HPV-positive cancer patients, the N-terminal vaccine killed two to three times more cancer cells than the other two vaccine models.
“This effect does not come from adding new ingredients or increasing the dose. It comes from a more intelligent presentation of the same components,” co-author of the study. Dr Jochen Lorchthe director of medical oncology at Northwestern Medicine’s Head and Neck Cancer Program, said in a statement. statement.
“The immune system is sensitive to the geometry of molecules,” he said. “By optimizing the way we attach the antigen to the ANS, immune cells processed it more efficiently.”
For the future, co-author of the study Chad Mirkininventor of SNAs and director of Northwestern’s International Nanotechnology Institute, hopes the approach could help scientists redesign older vaccines that initially looked promising but failed.
“This approach is poised to change the way we formulate vaccines,” Mirkin said in the release. “We may have left out perfectly acceptable vaccine components simply because they were in the wrong configurations. We can go back and restructure them and turn them into powerful drugs.”
This article is for informational purposes only and is not intended to offer medical advice.
Hwang, J. et al. (2026). The placement and orientation of E711-19 dictates the CD8+ T cell response in structurally defined spherical nucleic acid vaccines. Scientific advances, 12(7). https://doi.org/10.1126/sciadv.aec3876



