New evidence links heart disease to inflammation—and drugs can stop it

Doctors have been drilled for decades on the four big risks for heart disease, which kills more Americans every year than any other illness. The fearsome foursome: hypertension, smoking, high levels of “bad” LDL cholesterol and type 2 diabetes. Yet for just as long cardiologists have seen patients who have none of these problems die from heart ailments. And the heart specialists haven’t had the slightest idea why.
Up to a quarter of the people admitted to hospitals for heart attacks don’t have any of these four risk factors. Mysteriously, these “low-risk” heart disease patients actually have the worst outcomes. A 2023 analysis found that hospitalized acute coronary patients without any of the four hazards were 57 percent more likely to die compared with those who had at least one.
If the big known risk factors miss one in four patients, they still predict trouble as expected for the remaining three. That’s a good record. But it also means that of the roughly 920,000 Americans who die of cardiovascular disease every year, about 230,000 of them will have done so for no understandable reason.
On supporting science journalism
If you’re enjoying this article, consider supporting our award-winning journalism by subscribing. By purchasing a subscription you are helping to ensure the future of impactful stories about the discoveries and ideas shaping our world today.
This deadly puzzle has haunted cardiologist Paul Ridker for years. “I remember saying to myself that there must be some other fundamental determinant of heart disease,” says Ridker, who is director of the Center for Cardiovascular Disease Prevention at Brigham and Women’s Hospital in Boston.
Dozens of studies and clinical trials later, Ridker thinks he has found the missing piece. His work, along with that of other researchers, now suggests that chronic inflammation—a prolonged and body-damaging state of immune system activation—may be the hidden factor that accelerates cardiovascular problems to a dangerous and deadly state. When cholesterol builds up in the arteries, it shape-shifts into a sharp and jagged form the body no longer recognizes, provoking the immune system to wage war against it and blood vessels. It is a battle with no winners, and the wreckage it leaves behind ends in heart attacks and strokes.
Initially treated with skepticism, this idea now is becoming widely accepted by other scientists. Heart disease is “a disease of inflammation,” says Kathryn Moore, director of the Cardiovascular Research Center at New York University’s Grossman School of Medicine. And in the fall of 2025 the American College of Cardiology recommended that health-care providers routinely screen patients for inflammatory proteins.
If inflammation drives cardiovascular disease, then calming it might protect the organ beating in our chests.
The concept brings with it renewed hope for heart therapy. If inflammation drives cardiovascular disease, then calming it might protect the organ beating in our chests. In June 2023 the U.S. Food and Drug Administration approved a new use of an inexpensive, inflammation-reducing drug for an old disease—gout—to treat patients with heart disease. In a 2020 clinical trial, researchers showed this drug, colchicine, could reduce the risk of heart attacks, strokes, and other complications by a dramatic 31 percent. And this big decrease was mostly among patients already taking the standard cholesterol-lowering medications: statins.
But this treatment is not without controversy. Some recent studies of colchicine have not found protective effects, and many cardiologists are reluctant to use it. Ridker and other researchers are now testing different anti-inflammatory therapies with more precise modes of action. Although questions remain, many researchers believe this shift—seeing the vascular system not as a series of clogged pipes but as battlefields of inflammation—could transform public health and save millions of lives.
For many decades the dogma among doctors and scientists was that atherosclerosis—the buildup of fatty substances, including cholesterol, in the arteries—was a passive, almost mechanical process, an inevitable by-product of aging. “We were taught that the plaque buildup in arteries was sort of like rust in a pipe,” says Jean-Claude Tardif, a cardiologist who directs the Research Center at the Montreal Heart Institute.
Yet there had been hints over the centuries that inflammation might play an active role in the process. In the mid-1850s German pathologist Rudolf Virchow peered through his microscope at diseased blood vessels and saw angry, inflamed tissue within the plaques. In 1913 Russian pathologist Nikolai Anichkov wrote that he fed rabbits a high-cholesterol diet and found their arteries teeming with white blood cells, now known to be key markers of inflammation. For the most part, researchers interpreted these findings as evidence that inflammation might develop in response to atherosclerosis.
But it was also possible that things worked the other way around. Inflammation is a complicated process. It’s the body’s built-in alarm system, activated when the immune system senses that something untoward is happening. The body recruits immune cell soldiers to the scene, which launch an attack against any unwelcome intruders and cells they’ve infected. That’s why your throat gets red and swollen when you have the flu. Sometimes this alarm system becomes overzealous—fighting too hard or too long—and ends up harming the body’s healthy tissues in the process.
In the mid-1990s Ridker and his colleagues showed this hyperactivity could indeed be a cause. They published a landmark paper based on data collected as part of the Physicians’ Health Study, which had been gathering health data from male doctors since 1982. The researchers looked at blood levels of C-reactive protein (CRP), a molecular flare signaling inflammation, in 543 doctors who later had a heart attack, stroke or severe blood clot in a vein. The researchers compared these measurements with the levels of CRP in 543 doctors who did not go on to have a cardiac event. Compared with men who had the lowest CRP levels, those with the highest were three times as likely as to have a heart attack and twice as likely to have a stroke.
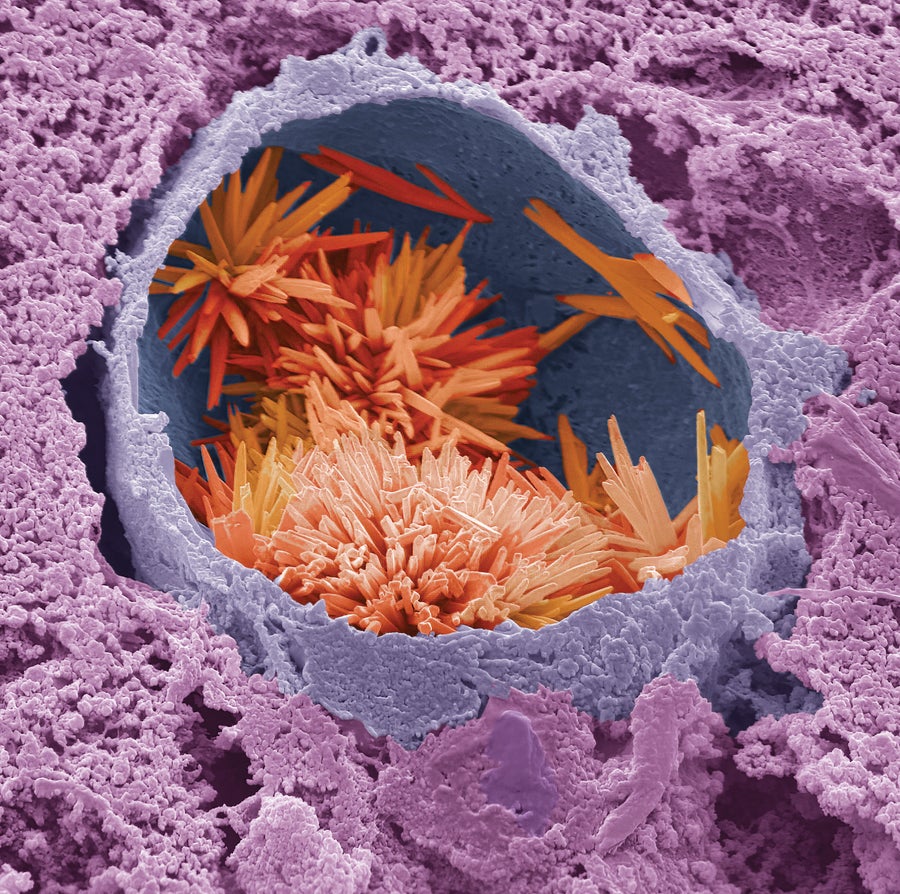
Cholesterol, a fatty compound produced primarily in the liver (above), can form jagged, needlelike crystals that tear artery walls and trigger an inflammatory reaction, blocking the flow of blood.
Steve Gschmeissner/Science Source
Several years later Ridker’s team studied nearly 28,000 women participating in the Women’s Health Study. The women were followed for an average of eight years. Those with the highest levels of CRP were 2.3 times more likely to have a heart attack or other serious cardiovascular problem than were women with the lowest levels.
These findings implied that inflammation wasn’t a side effect of heart disease. It was a lethal accomplice. In 1999 University of Washington pathologist Russell Ross wrote in the New England Journal of Medicine that “atherosclerosis is clearly an inflammatory disease.” Since then, other studies have confirmed Ridker’s findings. “Tons of studies involving hundreds of thousands of patients invariably have shown that the higher your inflammatory markers in blood, the higher the risk of clinical events,” Tardif says.
At that time, Ridker was also studying cholesterol-lowering statin drugs. These drugs are prescribed primarily for their ability to lower levels of LDL cholesterol, a known driver of heart disease. LDL, or low-density lipoprotein, is a molecule that carries cholesterol to tissues that need it—it helps to build cell membranes, among other crucial functions—but when there’s too much, it can seep into arteries and clog them.
Surprisingly, Ridker’s work started to suggest that statin drugs didn’t just reduce cholesterol—they damped inflammation, too. In a 2001 study, he and his colleagues showed that pravastatin, sold under the brand name Pravachol, reduced levels of CRP by 17 percent after 24 weeks. And in a 2005 study published in the New England Journal of Medicine, Ridker and his co-workers reported that regardless of their LDL cholesterol levels, people who took statins and had low CRP levels were less likely to have heart attacks or die from heart-related causes than people who took statins but had high CRP levels.
Ultimately the hope is not merely to prevent heart attacks and strokes but to stop atherosclerosis and heart disease before they start.
That discovery raised a radical question: Could people with healthy cholesterol levels—who typically would not be prescribed statins—still benefit from the drugs if their degree of inflammation was high?
Ridker decided to find out. He and his team designed a clinical trial to see whether people with low LDL but high CRP would benefit from statin therapy. They did—and markedly so. Published in 2008 and known as JUPITER, this trial enrolled nearly 18,000 participants with low LDL and high CRP to receive either a statin or a placebo. The trial was stopped early, after following people for an average of about two years, because the statins proved so effective. Those who took them were 54 percent less likely to have a heart attack and 48 percent less likely to have a stroke. A follow-up analysis of JUPITER patients showed that people most strongly protected from heart trouble were those who had lowered both their cholesterol and their CRP levels on the drug.
At the time, many doctors scoffed at the notion that cholesterol-lowering drugs could do anything useful for healthy people who already had low cholesterol. “They said, ‘These people don’t have risk,’” Ridker recalls. But the statins worked—and suddenly the old map of heart disease no longer made sense.
At first glance the JUPITER findings suggest that cholesterol’s true role in heart disease might be a small one. But the opposite is true: cholesterol, it turns out, is the match that lights the fire of vascular inflammation. A match in the shape of a crystal.
Ordinarily cholesterol is a waxy molecular blob. But when cholesterol accumulates and becomes highly concentrated in arteries, it can take on a crystalline shape, much the way sugar crystallizes in a concentrated syrup.
The crystals have been difficult for laboratory scientists to see. When pathologists are preparing biological tissue to be viewed under a microscope, they typically treat their sample with alcohol as part of the process of stiffening it so it can be thinly sliced and put on a slide. But alcohol dissolves cholesterol crystals, so pathologists rarely saw them when they studied slides of human and animal blood vessels. Instead they saw empty spaces that the crystals had left behind. And at any rate, researchers believed these crystals were harmless; some even postulated they arose primarily after death.
George Abela, chief of the division of cardiology at Michigan State University, had questions about these crystals. He knew that heart attacks and strokes often happened after arterial plaques ruptured, and he wondered whether cholesterol crystals might play a role, tearing the plaques. One day in 2004 he asked a colleague whether cholesterol might expand when it becomes crystalline, much as water expands when it forms ice. If it did, he surmised, then cholesterol could strain and harm arterial walls.
Abela recalls his colleague’s reply. “His response was, ‘No, the only [thing] on this planet that expands with crystallization is water,’” Abela says. That made sense to Abela. But he did go on to ask whether anyone had ever actually double-checked.
“Of course,” he says, “nobody had.”
Abela then had to see for himself. He bought cholesterol powder (which is used for various lab experiments), melted it into liquid form, and then let the cholesterol molecules crystallize. What he discovered shocked him: When it crystallized, cholesterol took up 45 percent more space than it did as a liquid. It did indeed expand. Moreover, the crystals had sharp, needlelike edges, perfectly shaped to tear artery walls. In a groundbreaking 2009 paper, Abela and his colleagues compared the coronary arteries of patients who had died of heart disease with those of people who had died for other reasons. They found that all the patients with heart disease had blood vessels that had been perforated by cholesterol crystals. “They’re like little knives cutting their way through the surfaces of the artery,” Abela explains. “And there are thousands of them.”
Crystals do even more damage than that. A healthy human body does not make crystalline structures, so our immune systems react to them as dangerous alarm signals and target them for attack. (This sequence of events happens in gout, too—the accumulation of crystal-shaped uric acid molecules leads to pain and inflammation.) Crystals “set off an inflammatory response to alert the body that these things are accumulating—‘we need to try to contain this and clear it,’” Moore says.
Next, white blood cells called monocytes race to the scene and change into macrophages. These are immune cells that engulf lipids and cholesterol deposits, including the sharp-edged cholesterol. Then, Moore explains, “they call in T cells and other immune cells to join the party, basically, and that is what causes the formation of the plaque,” a messy mixture of cholesterol, fats, immune cells and dead cell debris. The macrophages also produce inflammatory chemicals, including interleukin-1 beta (IL-1β), which further strengthen the immune attack and cause the plaque to worsen.
Normally the immune system fights, wins and then retreats. But inside an artery it doesn’t know how to quit. “For reasons that we don’t really understand, this inflammatory process gets going and doesn’t get turned off,” Moore says. It “can go on for decades.” Macrophages inside the arteries start to die, releasing their crystallized cholesterol contents. The plaque becomes increasingly unstable, and eventually it can rupture. A rupture exposes blood passing by to chemicals that cause it to clot. Those blood clots can travel through the body, causing heart attacks or strokes.
These discoveries and theories of action suggest that cholesterol is not the only bad guy in the story. The immune system’s overreaction to cholesterol crystals—the chronic inflammation it causes—is potentially far more dangerous.
After JUPITER highlighted the role of inflammation, Ridker wanted to see whether there was a remedy for it. Could cooling the inflammatory cycle help the heart? Because the cellular messenger IL-1β played such a key role in inflammation, he wondered whether blocking it might slow or stop the process, quieting the storm inside the arteries.
A few years after JUPITER was published, Ridker and his colleagues began recruiting people for a clinical trial that would end up convincing many other experts that inflammation-targeting therapies might be the next big cardiology frontier. In the trial, known as CANTOS and published in 2017, 10,000 heart disease patients with high CRP levels—most of whom were also on statins—were split into groups. Some were regularly injected with varying dosages of canakinumab, a monoclonal antibody that reduces levels of IL-1β in the body. Others were given a placebo.
After four years of the trial those who had received 150 milligrams of canakinumab were 15 percent less likely to die of heart disease or stroke compared with those who received a placebo. CANTOS “provided the first evidence in humans that blocking these inflammatory pathways would actually be beneficial,” Moore says. “It was considered a really big moment.”
Canakinumab did, however, cause side effects: those who took the drug were more likely than others to die from infections during the trial. The FDA rejected a 2018 bid to approve the use of the medication to treat heart disease.
But it turned out that there were other options. While CANTOS was underway, Stefan Mark Nidorf, a cardiologist affiliated with the Harry Perkins Institute of Medical Research in Perth, Australia, was pursuing a humbler approach. In a small clinical trial, he and his colleagues investigated the anti-inflammatory effects of colchicine, the old gout remedy. Nidorf’s team reasoned that because the medicine reduced inflammation, it might do the same for cardiac problems caused by cholesterol crystals. The drug, the researchers learned, did reduce the chances of coronary events in people with heart disease.
For better proof, Nidorf went bigger. In 2020 he and his team published the results of a much larger trial involving 5,500 heart disease patients. Those who took low daily doses of colchicine over an average of slightly more than two years were 31 percent less likely to experience a cardiac event compared with those who took a placebo. Almost all these people were also taking statins. That was important because it showed that colchicine could lower the chance of heart attacks and strokes among people who were already reducing their risk through cholesterol-lowering drugs.
“Atherosclerosis is a two-sided coin,” Nidorf says. “You’ve got to lower cholesterol because that drives the inflammatory process. But doing that alone isn’t enough—you’ve really got to add in the anti-inflammatory drug.” The fact that most people in the trial were already on statins suggested that colchicine had effects far above and beyond what statins could achieve.
In 2023, based in part on these findings, the FDA approved the use of colchicine to reduce the risk of heart attack, stroke, coronary revascularization, and cardiovascular death in patients with atherosclerotic disease or multiple risk factors for heart disease. That was followed two years later by the American College of Cardiology’s recommendation that health-care providers routinely screen patients’ CRP levels—a striking sign of how far the field has come.
But science rarely moves in straight lines. Since then, additional clinical trials involving colchicine have been published, and results have been mixed. In a clinical trial known as CLEAR SYNERGY, which was published in 2025 and involved 7,000 patients who had recently had a heart attack, researchers found that the use of colchicine for an average of three years did not reduce the participants’ risk of a coronary event.
“We were surprised, given the prior studies,” says the trial’s principal investigator, Sanjit Jolly, an interventional cardiologist at McMaster University in Ontario. The trial also raised concerns about colchicine’s gastrointestinal side effects. “Between 10 and 15 percent of patients get quite severe, quite significant diarrhea,” he adds. (Colchicine reduces inflammation by disrupting the formation of cellular structures called microtubules in immune cells. Microtubules are also essential to rapidly dividing cells that line the intestines, however, so people taking the drug may have GI problems.)
Therein lies a frequent difficulty with anti-inflammatory drugs. “When you’re affecting inflammation, you’re affecting the immune system,” says Gregg Stone, an interventional cardiologist at the Icahn School of Medicine at Mount Sinai. It’s often challenging to find a therapy “that hits the sweet spot of being effective without being too toxic.”
Perhaps because of the potential side effects, recent data suggest that cardiologists rarely prescribe colchicine. “I would say the general cardiology community has a lot of doubts right now,” Jolly says.
The CLEAR SYNERGY trial has exposed fault lines in the field. Ridker, Nidorf, and others argue that the trial had serious limitations. A major one was that it was conducted during the COVID-19 pandemic, when people might have been less likely to report heart-related issues to their doctors. It also focused on acute inflammation just after heart attacks, not the chronic inflammation of atherosclerosis. Colchicine may simply not offer benefits in that context, Nidorf says.
But other researchers say CLEAR SYNERGY produced sound conclusions, and it means that more research on colchicine is needed. “If you do have very high CRP, it’s probably a reasonable drug to try, but I just wouldn’t use it in everybody,” Stone says.
There are leads about different therapies. Several multinational clinical trials are underway to test the effects of inhibitors of interleukin-6, another of the immune system’s chemical messengers, on heart disease outcomes. IL-6 signaling drives a number of harmful vascular processes, and it’s an important link between IL-1β and CRP activity. It could be more closely tied to cardiovascular risks, so researchers hope IL-6 blockers will be more effective than other drugs. Some trials are being funded and overseen by large pharmaceutical companies, including Novo Nordisk and Novartis, but no results have been published yet.
Ultimately the hope is not merely to prevent heart attacks and strokes but to stop atherosclerosis and heart disease before they start. To that end, Tardif recently started a clinical trial involving 10,000 patients with type 2 diabetes to see whether colchicine can prevent heart disease in such people. Diabetes is a prime risk factor for cardiac trouble. “We’re trying to show that reducing inflammation very early in the process is going to help,” he says. At N.Y.U., Moore’s lab is trying to answer a related question: Is it possible to find drugs that can not only help to stop the immune fire but also repair the damage it’s already done?
Ridker thinks much of that is possible. After decades of skepticism, he is enjoying seeing his once radical idea go mainstream. The change “is very exciting,” he says. “Twenty years ago people thought we were just nuts.”



