Scientists create exotic new forms of ice never before seen on Earth

Chances are that all your encounters with frozen water – walking through muddy streets in winter, or treating yourself to cold lemonades in summer – have been limited to a structural form of ice, nicknamed Ih, the h referring to the hexagonal nature of its crystal lattice. But ice cream has much more to offer than that.
For more than a century, scientists have worked to push ice into extreme conditions, gradually creating more and more exotic structures. They have created more than 20 crystal forms to date, none of which we are likely to experience in our lifetime.
“Water is a beautiful, elegant system that constantly shows new and remarkable behavior,” says Ashkan Salamat, a physical chemist at the University of Nevada, Las Vegas. “For something so simple, it has a nice complexity to it.”
On supporting science journalism
If you enjoy this article, please consider supporting our award-winning journalism by subscription. By purchasing a subscription, you are helping to ensure the future of impactful stories about the discoveries and ideas shaping our world today.
At the heart of all these exotic ices – and our more mundane ices, as well as water and steam – is the same molecule: H2O, an oxygen atom flanked by hydrogen atoms forming an angle of 104.5 degrees. In each variety of ice cream, H2O molecules interact, with weak connections called hydrogen bonds forming between an oxygen atom and a hydrogen atom in separate molecules. Different arrangements of these hydrogen bonds can shape the crystal structure of ice into various configurations, ranging from a hexagonal prism to a cubic lattice to less familiar lattice systems such as rhombohedral and tetragonal.
Hydrogen bonds between water molecules are extremely sensitive to changes in temperature and pressure, Salamat explains, giving water what he calls “quantum behavior.” Molecules are forced to establish radically different relationships with each other at certain thresholds of these conditions. So he and other scientists conjure up obscure recipes—crushing water at 3,000 times atmospheric pressure, for example, or chilling it (with a splash of potassium hydroxide) to -200 degrees Celsius (-330 degrees Fahrenheit) for a week—all in the search for new forms of ice.
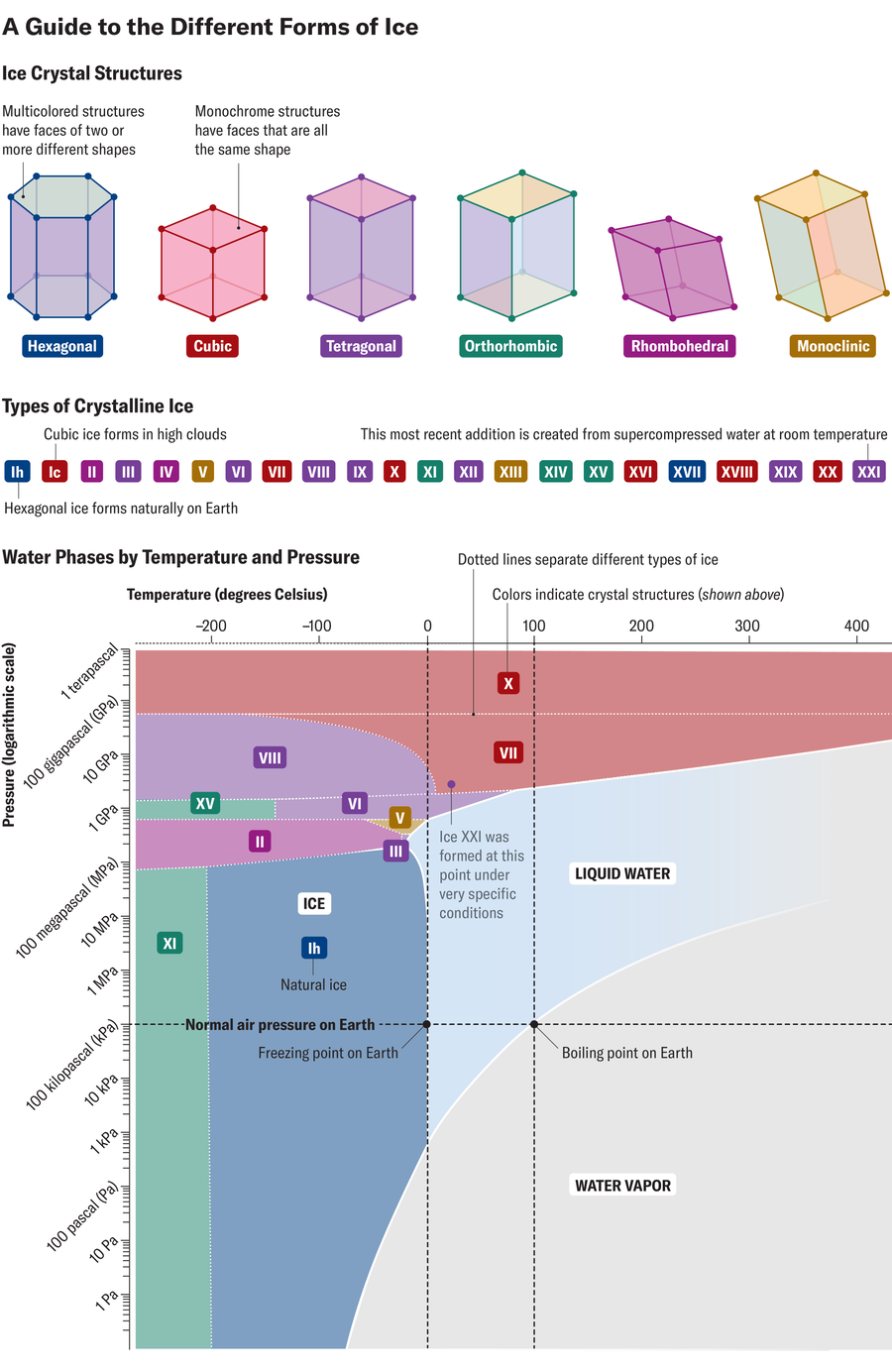
The latest frozen discovery is ice XXI, announced in Natural materials. (Salamat was not involved in this work, although his team published the discovery of a new transition phase dubbed ice VIIt in 2022.) Ice XXI is an ephemeral, blocky crystalline structure that grows from supercompressed water: Scientists were only able to see it using an extremely powerful X-ray free-electron laser that essentially functions like a high-speed camera.
“Observing things at a very, very fast pace allows us to observe weird and wonderful phenomena,” says Salamat, calling the laser “an incredibly exciting new toy.” The laser allows researchers to spot exotic ices that only exist briefly, by introducing time as a variable alongside temperature and pressure.
Although they don’t exist naturally on Earth, some of these strange ice forms can form on other worlds: deep within Neptune, trapped in a distant moon, or somewhere even stranger. But for Salamat, the laboratory can be just as exotic. “There are still new and exciting things we can discover,” he says.
It’s time to defend science
If you enjoyed this article, I would like to ask for your support. Scientific American has been defending science and industry for 180 years, and we are currently experiencing perhaps the most critical moment in these two centuries of history.
I was a Scientific American subscriber since the age of 12, and it helped shape the way I see the world. SciAm always educates and delights me, and inspires a sense of respect for our vast and magnificent universe. I hope this is the case for you too.
If you subscribe to Scientific Americanyou help ensure our coverage centers on meaningful research and discoveries; that we have the resources to account for decisions that threaten laboratories across the United States; and that we support budding and working scientists at a time when the value of science itself too often goes unrecognized.
In exchange, you receive essential information, captivating podcasts, brilliant infographics, newsletters not to be missed, unmissable videos, stimulating games and the best writings and reports from the scientific world. You can even offer a subscription to someone.
There has never been a more important time for us to stand up and show why science matters. I hope you will support us in this mission.



