They’re in a race to save their children with a rare disease. A new drug has given them hope.

Like other children with Hunter syndrome, Cole lacks an enzyme needed to break down certain molecules. Over time, the toxins build up and the genetic disease ravages children’s organs, including their hearts – and in many cases, their brains, leading to dementia-like symptoms. This disease, also called mucopolysaccharidosis type II, or MPS II, affects about 500 people in the United States, almost all of them boys.
Experts say the newly approved drug, an intravenous enzyme replacement therapy made by Denali Therapeutics, will be a game-changer, particularly because current standards of care only slow the physical aspects of the disease. Denali’s drug also targets cognitive decline.
The new drug will not reverse regressions that have already occurred. But it could extend children’s lives and prevent many symptoms from developing in those who receive it early.

“If we take a child very young and can treat them before they are damaged, the potential is now almost limitless,” said Dr. Joseph Muenzer of the Muenzer MPS Research and Treatment Center at the University of North Carolina at Chapel Hill, who sees children with Hunter syndrome and other rare mucopolysaccharide diseases.
“We don’t know how successful they will be in the future, but they will make a radically different outcome than they would have done otherwise,” he said.
Before he began to regress, Stephens’ son, Cole, was learning to read and could speak in complete sentences. As the illness took hold, speaking became difficult: he could string a few words together, then utter a single word – “Mom” – before becoming completely nonverbal. Although he is now a teenager, he is more like a 3-year-old developmentally, Stephens said.
The FDA’s approval of the drug Denali came as a welcome surprise not only to families of children with Hunter syndrome, but also to the rare disease community as a whole. In recent months, the FDA has been criticized for rejecting a series of promising treatments for rare diseases, prompting patient advocates to hold a mock funeral with a coffin outside the agency’s headquarters and sparking an investigation by Sen. Ron Johnson, R-Wis., who, before Denali’s approval, accused the FDA of “making excuses to say no” to treatments.
In an email to NBC News, the FDA said the number of approvals and denials under this administration “is consistent with historical data over the past decade.” He highlighted a statement from FDA Commissioner Dr. Marty Makary, who called Denali’s approval “an important day for children and their families battling Hunter syndrome.” He added: “We will continue to do everything we can to accelerate treatments for rare diseases. »
Those who have seen children suffer from Hunter syndrome and other rare diseases hope this will be the case, including Muenzer, who was one of the principal investigators of the Denali trial.
“This is terrible unrest,” Muenzer said. “Just because they are a minority doesn’t mean we should ignore them.”
Denali’s drug, called Avlayah, is the first FDA-approved treatment in the United States in 20 years for Hunter syndrome – and the first to cross the blood-brain barrier, allowing it to halt the neurological complications of the disease.
Experts like Muenzer believe Avlayah has the potential to extend life expectancy based on promising data from a clinical trial that showed that after 24 weeks, levels of a key biomarker in the spinal fluid associated with the disease were so reduced that 93% of pediatric participants had levels comparable to individuals without Hunter syndrome.
For many families of children with the disease, the drug’s approval meant more than an exciting new treatment. It also meant recognition of the value of their children.
In New Berlin, Wisconsin, 6-year-old Roran Jaskulski was diagnosed with Hunter syndrome when he was 4 years old. He has always been nonverbal, and his mother, Kylie Jaskulski, said she worries that because Roran can’t speak, people who don’t know him might wonder if he has anything to offer others — a heartbreaking description of her energetic, affectionate child who loves to cuddle as he falls asleep and excitedly runs to school each day.
“He brings so much joy,” Jaskulski said. “He brings happiness and peace to every person he interacts with.”
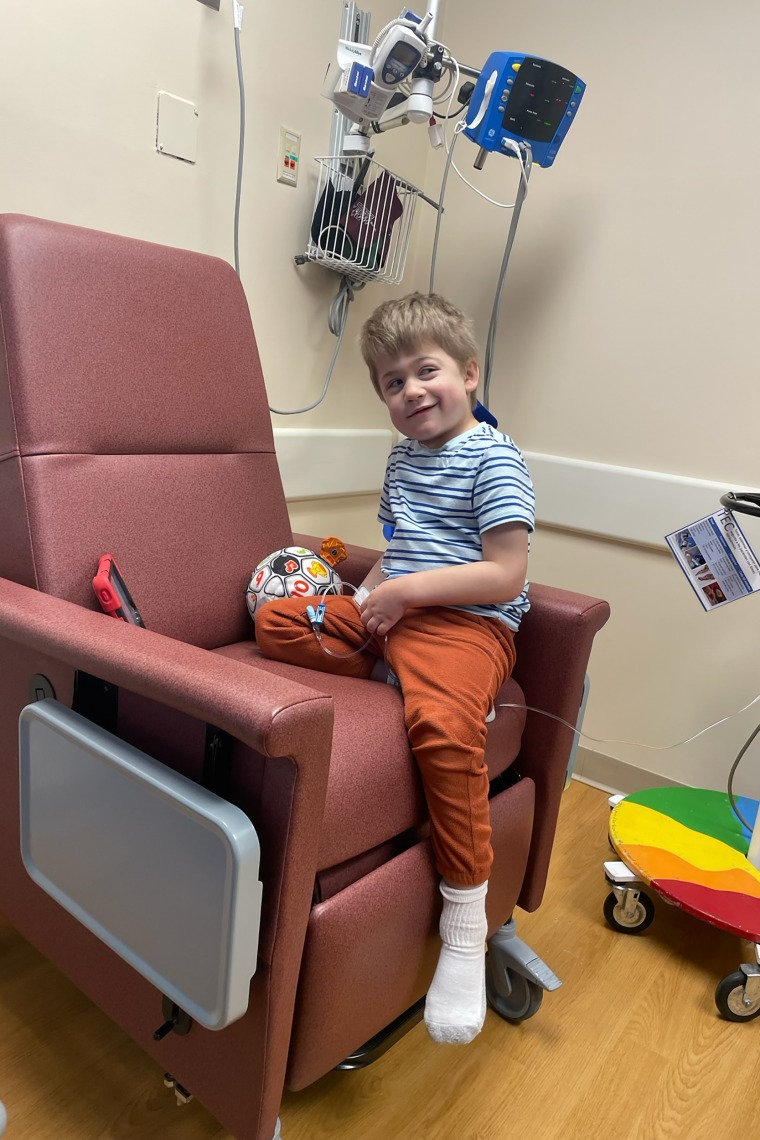
Jaskulski said at first receiving Roran’s diagnosis was the worst day of his life. But as time passed, she realized that her powerlessness to prevent her son’s deterioration was even more tortuous.
If her insurance approves the Denali drug for Roran, she said, “maybe I won’t have to stand by and watch.”
Like other children with Hunter syndrome, Roran receives a weekly infusion of the current standard treatment, a drug called Elaprase approved by the FDA in 2006. The infusions help stabilize his physical decline, but not entirely: In recent months, Jaskulski noticed that Roran was developing weakness and pain on the left side of his body, affecting his gait. He also suffers from slight hearing loss.
A patchwork of states, from California to Rhode Island, include Hunter syndrome in newborn screenings, and more states are expected to begin testing for it at birth in the future. That means children will have a better chance of preventing cognitive impairment if they take the new drug earlier, doctors say.
Most children with Hunter syndrome suffer from the severe form of the disease, with a life expectancy of between 10 and 20 years. People with the non-neurological form of Hunter syndrome, which does not significantly affect the brain, can live into adulthood, although they still face progressive physical problems, primarily with the airways and heart.

The prospect of this new treatment is exciting for families of children at both ends of the disease. In Newkirk, Oklahoma, Kashton Estes, Christina Coldwell’s 3-year-old grandson, has Hunter syndrome without cognitive problems, and he receives Elaprase infusions, which he calls “going to get his Spider-Man juice.” The treatment was successful: Coldwell said the little boy “talks like a storm,” runs, jumps and rides a bike.
Still, she can’t wait for Kashton to take the new drug Denali. Two of his cousins also have the genetic disorder, Coldwell said, and the family wants to avoid future health problems in all three children.
“We are not asking for much: just give us the medicines we need to keep our children alive,” she said.
Asked about insurance coverage for its drug — which has a list price of $5,200 per 150-milligram vial — Denali Therapeutics said it had “constructive” discussions with payers and said allowing families with Hunter syndrome to have timely access to Avlayah was a “top priority.” The company also said it is looking to expand the drug’s clinical evidence in young adults, as at the moment it is only indicated in pediatric patients. Denali also said it plans to use its blood-brain barrier technology for a wide range of other conditions, including other neurodegenerative diseases.
Stephens, the mother of 15-year-old Cole, can’t wait to start her son on the medication. She dedicated her life to helping not only Cole, but others like him: in 2022, she became executive director of the Muenzer MPS Research and Treatment Center at UNC-Chapel Hill.
When news broke last week that the FDA had granted accelerated approval to the new drug, Stephens ran into the treatment center to tell patients and staff. Everyone hugged and cried.
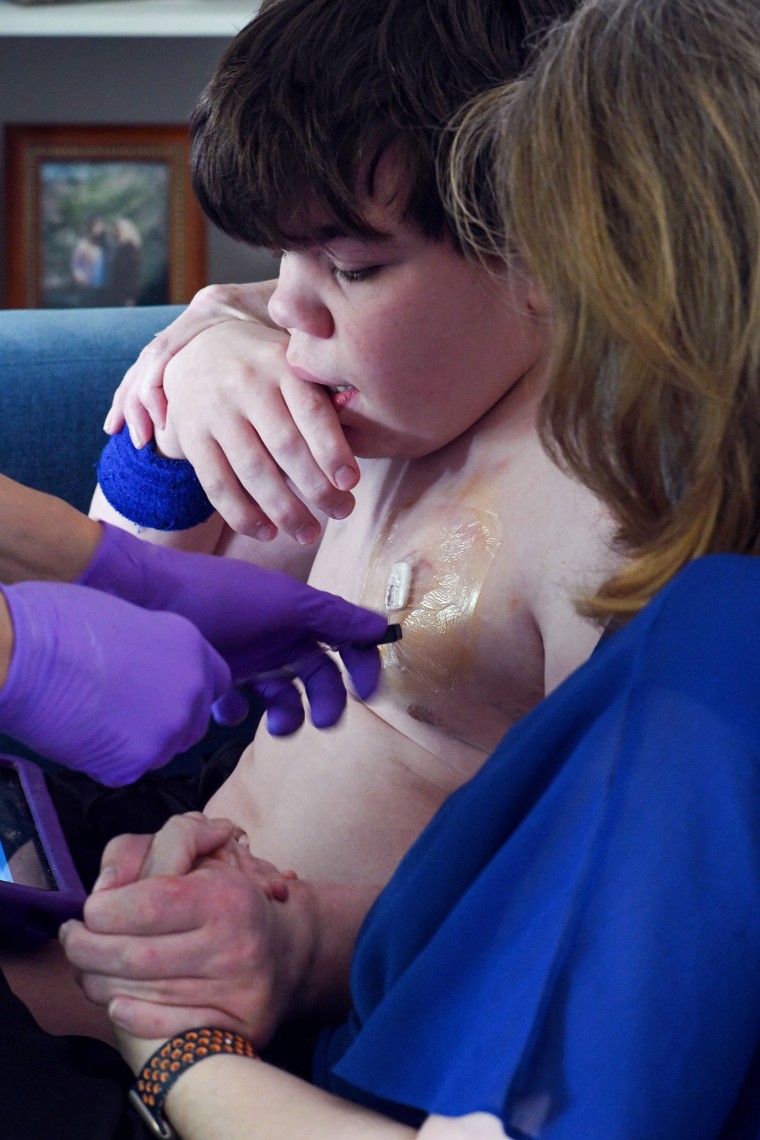
Stephens knows that Denali’s medicine can’t undo the regressions Cole already has. This will not allow him to go to college or enter the job market. But she said she’s still incredibly grateful for it.
“I hope it stays stable,” Stephens said. “Stable with progressive disease is a victory. »



